Advertisements
Advertisements
Question
Write information about the given atomic numbers in the table. 10, 20, 7.
| Atomic Number | Electronic configuration | Group | Period | Element |
| 10 | ||||
| 20 | ||||
| 7 |
Advertisements
Solution
| Atomic Number | Electronic configuration | Group | Period | Element |
| 10 | 2, 8 | 18 | 2 | Neon (Ne) |
| 20 | 2, 8, 8, 2 | 2 | 4 | Calcium (Ca) |
| 7 | 2, 5 | 15 | 2 | Nitrogen (N) |
APPEARS IN
RELATED QUESTIONS
Answer the following in respect of element `31/15 P`
Give its electronic configuration
Answer the following in respect of element `31/15 P `
Give its formula with chlorine?
Give the trends in atomic size on moving down the group.
Which is greater in size Fe2+ or Fe3+?
Arrange the following in increasing order of property indicated
I, I+, I- (atomic size)
Select the correct answer
Atomic radii of fluorine and neon in angstrom unit are
What happens to the atomic size of elements on moving from left to right in a period?
Supply the missing word from those in the brackets:
If an element has seven electrons in its outermost shell then it is likely to have the ______ (largest/ smallest) atomic size among all the elements in the same period.
The metals of Group 2 from top to bottom are Be, Mg, Ca, Sr and Ba. Which one of these elements will form ions most readily and why?
Arrange the following as per the instruction given in the bracket
Na, K, Li (Increasing atomic size)
On moving from left to right in a periodic table, the size of the atom _______.
Write an Explanation.
Atomic radius
Which of the following are the characteristics of isotopes of an element?
- Isotopes of an element have same atomic masses
- Isotopes of an element have same atomic number
- Isotopes of an element show same physical properties
- Isotopes of an element show same chemical properties
Elements have been arranged in the following sequence on the basis of their increasing atomic masses.
| F, | Na, | Mg, | Al, | Si, | P, | S, | Cl, | Ar, | K |
- Pick two sets of elements which have similar properties.
- The given sequence represents which law of classification of elements?
Arrange the following elements in increasing order of their atomic radii.
- Li, Be, F, N
- Cl, At, Br I
An element X (atomic number 17) reacts with an element Y (atomic number 20) to form a divalent halide.
- Where in the periodic table are elements X and Y placed?
- Classify X and Y as metal (s), non-metal (s) or metalloid (s)
- What will be the nature of oxide of element Y? Identify the nature of bonding in the compound formed
- Draw the electron dot structure of the divalent halide
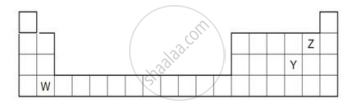
The diagram below shows part of the periodic table.
- Which elements would react together to form covalent compounds?
- Between the two elements W and Z, which will have a bigger atomic radius? Why?
Arrange the following as per the instruction given in the bracket:
Carbon, Fluorine, Beryllium (decreasing order of atomic size).
