Advertisements
Advertisements
Question
An element X (atomic number 17) reacts with an element Y (atomic number 20) to form a divalent halide.
- Where in the periodic table are elements X and Y placed?
- Classify X and Y as metal (s), non-metal (s) or metalloid (s)
- What will be the nature of oxide of element Y? Identify the nature of bonding in the compound formed
- Draw the electron dot structure of the divalent halide
Answer in Brief
Advertisements
Solution
- Element X is chlorine, it is placed at 17th group and 3rd period in the periodic table. Element Y is calcium, it is placed at the 2nd group and 4th period in the periodic table.
- Element X is a non-metal and element Y is a metal.
- Element Y is metal and thus, the oxide of this metal will be basic in nature. The chemical formula for calcium oxide is CaO and the bonding in the compound is ionic in nature.
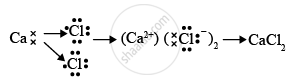
shaalaa.com
Is there an error in this question or solution?
