Advertisements
Advertisements
Question
The changes in the properties of elements on moving from left to right across a period of the Periodic Table. For the property, choose the correct answer.
The atomic size:
Options
Decreases
Increases
Remains the same
Sometimes increases and sometimes decreases
Advertisements
Solution
Decreases
APPEARS IN
RELATED QUESTIONS
Answer the following in respect of element `31/15 P `
Is it a reducing agent or oxidizing agent?
What do you understand by atomic size? State its unit?
Give the trends in atomic size on moving down the group.
Size of atom progressively becomes smaller when we move from sodium (Na) to chlorine (CI) in the third period of the periodic table?
_______ is the distance between the nucleus of the atom and its outermost shell.
Nanometer unit is used to measure atomic radius.
The size of an atom depends on the number of valence electrons.
Which of the following gives the correct increasing order of the atomic radii of O, F, and N?
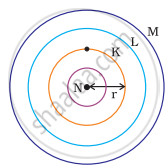
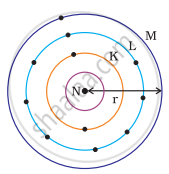
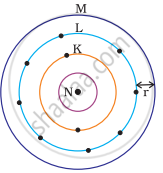
Which one of the following depict the correct representation of atomic radius(r) of an atom?
|
(i) |
(ii) |
|
(iii) |
(iv) |
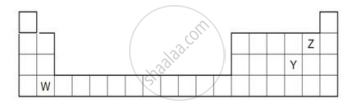
The diagram below shows part of the periodic table.
- Which elements would react together to form covalent compounds?
- Between the two elements W and Z, which will have a bigger atomic radius? Why?