Advertisements
Advertisements
Question
Write the formula of the product formed when the element A (atomic number 19) combines with the element B (atomic number 17). Draw its electronic dot structure. What is the nature of the bond formed?
Answer in Brief
Advertisements
Solution
Element A has atomic number 19 so, it is potassium. Element B has atomic number 17 so, it is chlorine. As the valency of potassium is 1 and the valency of chlorine is also 1, the formula of the compound formed would be KCl or AB.
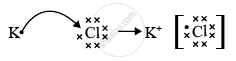
The nature of the bond between potassium and chlorine in KCl is an ionic bond.
shaalaa.com
Is there an error in this question or solution?
