Advertisements
Advertisements
प्रश्न
Explain thermodynamics of the adiabatic process.
Advertisements
उत्तर
- An adiabatic process is a process during which there is no transfer of heat from or to the system.
- For adiabatic change, Q = 0.
∴ ΔU + W = 0
∴ ΔU = –W - This implies, for adiabatic expansion, W is positive and ΔU is negative i.e. when work is done by the system adiabatically its internal energy decreases.
- Similarly, when a system is compressed adiabatically, W is negative and ΔU is positive i.e., the internal energy of the system increases in an adiabatic compression.
- Heat transfer to and from the system is prevented by either perfectly insulating the system from its environment or by carrying out the change rapidly so that there is no time for any exchange of heat.
- The temperature of the system changes, i.e., ΔT ≠ 0
- Usually, it is a sudden change and the system does not find any time to exchange heat with its environment.
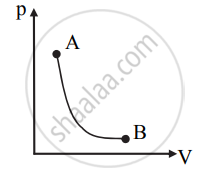
- p-V diagram for the adiabatic process is as shown below:
APPEARS IN
संबंधित प्रश्न
Give an example of some familiar process in which heat is added to an object, without changing its temperature.
Heating a gas in a constant volume container is an example of which process?
Draw a p-V diagram showing negative work with varying pressure.
Explain graphically (i) positive work with varying pressure, (ii) negative work with varying pressure, and (iii) positive work at constant pressure.
When food is cooked in a vessel by keeping the lid closed, after some time the steam pushes the lid outward. By considering the steam as a thermodynamic system, then in the cooking process
When you exercise in the morning, by considering your body as a thermodynamic system, which of the following is true?
In an isochoric process, we have ____________.
Give an expression for work done in an isothermal process.
Apply first law for an isothermal process.
Apply first law for an adiabatic process.
Give the equation of state for an adiabatic process.
Draw the PV diagram for the isothermal process.
Draw the PV diagram for the adiabatic process.
What is meant by a reversible and irreversible processes?
Can the given heat energy be completely converted to work in a cyclic process? If not, when can the heat can completely converted to work?
Derive the work done in an isothermal process.
Derive the work done in an adiabatic process.
Explain in detail the isochoric process.
What are the limitations of the first law of thermodynamics?
In a petrol engine, (internal combustion engine) air at atmospheric pressure and temperature of 20°C is compressed in the cylinder by the piston to `1/8` of its original volume. Calculate the temperature of the compressed air. (For air γ = 1.4)
Consider the following cyclic process consist of isotherm, isochoric and isobar which is given in the figure.

Draw the same cyclic process qualitatively in the V-T diagram where T is taken along the x-direction and V is taken along the y-direction. Analyze the nature of heat exchange in each process.
One mole of an ideal gas with `gamma` = 1.4 is adiabatically compressed so that its temperature rises from 27° C to 47° C. The change in the internal energy of the gas is (R = 8.3 J/mol.K) ____________.
In which of the following processes, beat is neither absorbed nor released by a system?
Consider P-V diagram for an ideal gas shown in figure.

Out of the following diagrams (figure), which represents the T-P diagram?
 (i) |
 (ii) |
 (iii) |
 (iv) |
Give any two types of a thermodynamic process.
In the figure shown here, the work done in the process ACBA is ______.

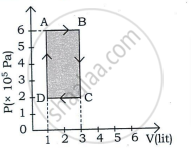
An ideal gas is taken through a cyclic process ABCDA as shown in figure. The net work done by the gas during the cycle is ______.
Explain how can a gas be expanded at constant temperature.
