Advertisements
Advertisements
प्रश्न
An organic compound having the molecular formula C3H6O can exist in the form of two isomers A and B having different functional groups. The isomer A is a liquid which is used as a solvent for nail polish. The isomer B is also a liquid. An aqueous solution of one of the lower homologues of B is used for preserving biological specimens in the laboratory
(a) What is compound A?
(b) Write the electron-dot structure of A.
(c) What is compound B?
(d) Write the electron-dot structure of B.
(e) Name the lower homologue of compound B which is used in preserving biological specimens.
Advertisements
उत्तर
(a) The isomer A is propanone (acetone).
(b) The electron-dot structure of propanone is::
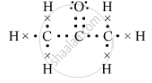
(c) The isomer B is propanal.
(d)The electron-dot structure of propanal is:
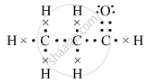
(e) Methanal or formaldehyde is the lower homologue of compound B, which is used to preserve biological specimens.
APPEARS IN
संबंधित प्रश्न
State any four characteristics of a homologous series
What is meant by homologous series of carbon compounds?
Complete the following table which relates to the homologous series of hydrocarbons.
|
General Formula |
IUPAC name of the homologous series | Characteristic bond type | IUPAC name of the first member of the series |
| `C_nH_(2n-2)` | (A)________ | (B)______ | (C)______ |
| C_nH_(2n+1) | (B)________ | (E)______ | (F)______ |
By how many carbon atoms and hydrogen atoms do any two adjacent homologues differ?
Write the names and formulae for the first three members of the homologous series for chloroalkanes.
The molecular formula of a homologue of butane is:
(a) C4H8
(b) C3H6
(c) C4H6
(d) C3H8
The number of carbon atoms present in the molecule of fifth member of the homologous series of alkynes is:
(a) four
(b) five
(c) six
(d) seven
A colourless organic liquid X of molecular formula C2H4O2 turns blue litmus to red. Another colourless organic liquid Y of molecular formula C3H6O has no action on any litmus but it is used as a nail polish remover. A yet another colourless organic liquid Z of molecular formula C2H6O has also no action on litmus but it is used in tincture of iodine.
(a) Name the liquid X. To which homologous series does it belong? Give the name of another member of this homologous series.
(b) Name the liquid Y. To which homologous series does it belong? Write the name of another member of this homologous series.
(c) Can you name an organic compound having the same molecular formula as liquid Y but which belongs to a different homologous series? What is this homologous series?
(d) Name the liquid Z. To which homologous series does it belong? Write the name of another member of this homologous series.
Give the abbreviated formula of the third member of the alcohol.
Give the structure of the second member of the alcohol group.
Give the names of the first four members of the homologous series of
alkynes.
What is the difference in the molecular formula of any two adjacent homologues:
(i) In terms of molecular mass
(ii) In terms of number and kind of atoms per molecule?
Assertion (A): In a homologous series of alcohols, the formula for the second member is C2H5OH and the third member is C3H7OH.
Reason (R): The difference between the molecular masses of the two consecutive members of a homologous series is 144.
The unsaturated hydrocarbons containing a carbon-carbon double bond are called _______.
Cyclohexane : Cyclic hydrocarbon : : Isobutylene : _______
Saturated hydrocarbon : Single bond : : Unsaturated hydrocarbon : _______
Find the odd one out and give its explanation.
Complete the following table for homologous series of alcohols.
| Name | Molecular formula | Condensed structural formula | Number of carbon atom | Number of -CH2- units | Boiling point °C |
| Methanol | CH4O | CH3-OH | 1 | 1 | 63 |
| Ethanol | C2H6O | CH3–CH2-OH | 2 | 2 | 78 |
| Propanol | C3H8O | CH3–CH2–CH2-OH | ______ | ______ | 97 |
| Butanol | C4H10O | CH3–CH2–CH2–CH2–OH | ______ | ______ | 118 |
The first member of alkyne homologous series is
