Advertisements
Advertisements
प्रश्न
 .
.
Advertisements
उत्तर
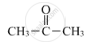
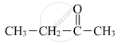
APPEARS IN
संबंधित प्रश्न
Write the molecular formula of the 2nd and 3rd member of the homologous series where the first member is ethyne.
Fill in the following blank with suitable word:
Ethene and ethyne are examples of ..... hydrocarbons.
What is a homologous series? Explain with an example.
Assertion (A): In a homologous series of alcohols, the formula for the second member is C2H5OH and the third member is C3H7OH.
Reason (R): The difference between the molecular masses of the two consecutive members of a homologous series is 144.
Complete the correlation:
Alkene : C = C :: Alkyne: _______.
While going in an increasing order there is a rise in the molecular mass of the consecutive members of the homologous series by _______.
There are different general molecular formula for all members of the homologous series.
Complete the following table for homologous series of alcohols.
| Name | Molecular formula | Condensed structural formula | Number of carbon atom | Number of -CH2- units | Boiling point °C |
| Methanol | CH4O | CH3-OH | 1 | 1 | 63 |
| Ethanol | C2H6O | CH3–CH2-OH | 2 | 2 | 78 |
| Propanol | C3H8O | CH3–CH2–CH2-OH | ______ | ______ | 97 |
| Butanol | C4H10O | CH3–CH2–CH2–CH2–OH | ______ | ______ | 118 |
Successive members of a homologous series vary by how many atomic mass unit?
The first member of alkyne homologous series is
