Advertisements
Advertisements
प्रश्न
What is a homologous series? Explain with an example.
Advertisements
उत्तर
- A group of compounds having similar structures and chemical properties but their successive compounds differ by a CH2 group is called a homologous series.
- Example: Alkanes have similar structures and similar chemical properties, so they can be grouped together to form a homologous series. The general formula of the homologous series of alkanes is CnH2n+2, where n is the number of carbon atoms in each molecule.
APPEARS IN
संबंधित प्रश्न
Write the next homologue of the following: C4H6
Write the name and formula of the 2nd member of homologous series having general formula CnH2n + 2.
Give the structural formula for 1, 2-dichloroethane
Define a homologous series. Give the name and structural formula of one homologue of the following:
CH3OH
Succeeding members of a homologous series differ by ______.
Give the structure of the second member of the alcohol group.
Copy and complete the following table, which relates to the three homologous series of hydrocarbons:
| General formula | CnH2n | CnH2n-2 | CnH2n+2 |
| IUPAC name of the homologus series | |||
| Characteristic bond type | Single bonds | ||
| IUPAC name of the first member of the series | |||
| Type of reaction with chlorine | Addition |
Write the name and formula of the fourth member of the following homologous series:
Alkyne
What is a homologous series?
The general formula of Alkane is _________________
Assertion (A): In a homologous series of alcohols, the formula for the second member is C2H5OH and the third member is C3H7OH.
Reason (R): The difference between the molecular masses of the two consecutive members of a homologous series is 144.
The general molecular formula for the homologous series of alkynes is _______.
Saturated hydrocarbon : Single bond : : Unsaturated hydrocarbon : _______
Observe the structural formula and answer the following questions.
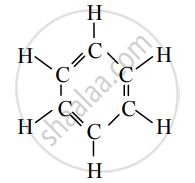
- Write the name of the given hydrocarbon.
- The given hydrocarbon is included in which type of hydrocarbon?
- What is the kind of compounds with the above characteristic structure called?
The first member of alkyne homologous series is
Study and complete the following table:
| Homologous series | Alkane | Alkyne |
| General formula | CnH2n+2 | 1. ______ |
| IUPAC name | 2. ______ | Ethyne |
| Common name | Marsh gas | 3. ______ |
A carbon compound ‘A’ having melting point 156K and boiling point 351K, with molecular formula C2H6O is soluble in water in all proportions.
- Identify ‘A’ and draw its electron dot structure.
- Give the molecular formulae of any two homologues of ‘A’.
Define Homologous series.
Consider the carbon compounds having following molecular formula:
(i) C3H6 (ii) C3H8 (iii) C4H6 (iv) C6H6 (v) C6H12
- State the number of double covalent bonds present in C3H6.
- Write the formula of first member of the homologous series to which the carbon compound C4H6 belongs.
- Which one of the above compounds forms a ring structure of carbon atoms?
- Identify, which of the above compounds, is a member of alkane series.
Consider the carbon compounds having following molecular formula:
(i) C3H6 (ii) C3H8 (iii) C4H6 (iv) C6H6 (v) C6H12
- State the number of double covalent bonds present in C3H8.
- Write the formula of first member of the homologous series to which the carbon compound C4H6 belongs.
- Which one of the above compounds forms a ring structure of carbon atoms?
- Identify, which of the above compounds, is a member of alkane series.
