Advertisements
Advertisements
प्रश्न
A carbon compound ‘A’ having melting point 156K and boiling point 351K, with molecular formula C2H6O is soluble in water in all proportions.
- Identify ‘A’ and draw its electron dot structure.
- Give the molecular formulae of any two homologues of ‘A’.
Advertisements
उत्तर
- Ethanol; C2H5OH
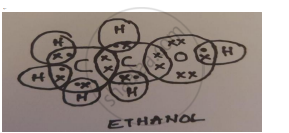
- CH3OH and C3H7OH are homologues of ethanol
OR
CH4O and C₃H₈O
APPEARS IN
संबंधित प्रश्न
Classify the following carbon compounds into two homologous series and name them.
C3H4, C3H6, C4H6, C4H8, C5H8, C5H10
Give the structural formula for 1, 2-dichloroethane
The number of carbon atoms present in the molecule of fifth member of the homologous series of alkynes is:
(a) four
(b) five
(c) six
(d) seven
Give the structure of the second member of the alcohol group.
Give the names and the structural formula of the first three members of the homologous series of alkanes.
Study the different conclusions drawn by students of a class on the basis of observations of preserved/available specimens of plants and animals.
I. Potato and sweet potato are analogous organs in plants.
II. Wings of insects and wings of birds are homologous organs in animals.
III. Wings of insects and wings of bats are analogous organs in animals.
IV. Thorns of citrus and tendrils of cucurbita are analogous organs in plants.
The correct conclusions are:
(A) I, and II
(B) II and IV
(C) I and III
(D) III and IV
Give the names of the first four members of the homologous series of
alkynes.
Write the name and formula of the fourth member of the following homologous series:
Alkyne
Cyclohexane : Cyclic hydrocarbon : : Isobutylene : _______
Name the third homologue of alcohols.
