Advertisements
Advertisements
प्रश्न
A carbon compound ‘A’ having melting point 156K and boiling point 351K, with molecular formula C2H6O is soluble in water in all proportions.
- Identify ‘A’ and draw its electron dot structure.
- Give the molecular formulae of any two homologues of ‘A’.
Advertisements
उत्तर
- Ethanol; C2H5OH
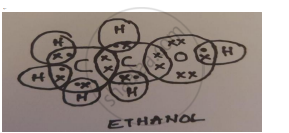
- CH3OH and C3H7OH are homologues of ethanol
OR
CH4O and C₃H₈O
APPEARS IN
संबंधित प्रश्न
Write the next homologue of the following: C4H6
Classify the following carbon compounds into two homologous series and name them.
C3H4, C3H6, C4H6, C4H8, C5H8, C5H10
Write the molecular formula of two consecutive members of homologous series of aldehydes. State which part of these compounds determines their
- physical and
- chemical properties
Give the molecular formula of one homologue of each of the following:
C2H6
The number of carbon atoms present in the molecule of fifth member of the homologous series of alkynes is:
(a) four
(b) five
(c) six
(d) seven
An organic compound having the molecular formula C3H6O can exist in the form of two isomers A and B having different functional groups. The isomer A is a liquid which is used as a solvent for nail polish. The isomer B is also a liquid. An aqueous solution of one of the lower homologues of B is used for preserving biological specimens in the laboratory
(a) What is compound A?
(b) Write the electron-dot structure of A.
(c) What is compound B?
(d) Write the electron-dot structure of B.
(e) Name the lower homologue of compound B which is used in preserving biological specimens.
Study the different conclusions drawn by students of a class on the basis of observations of preserved/available specimens of plants and animals.
I. Potato and sweet potato are analogous organs in plants.
II. Wings of insects and wings of birds are homologous organs in animals.
III. Wings of insects and wings of bats are analogous organs in animals.
IV. Thorns of citrus and tendrils of cucurbita are analogous organs in plants.
The correct conclusions are:
(A) I, and II
(B) II and IV
(C) I and III
(D) III and IV
Give the names of the first four members of the homologous series of
alkynes.
The first member of alkyne homologous series is
