Advertisements
Advertisements
प्रश्न
What is the difference in the molecular formula of any two adjacent homologues:
(i) In terms of molecular mass
(ii) In terms of number and kind of atoms per molecule?
Advertisements
उत्तर
The difference in the molecular formula of any two adjacent homologues is
(i) It differs by 14 a.m.u in terms of molecular mass.
(ii) It differs by three atoms. The kind of atoms it differs is one carbon and two hydrogens.
संबंधित प्रश्न
What is meant by homologous series of carbon compounds?
Write the name and formula of the 2nd member of homologous series having general formula CnH2n.
Fill in the following blank with suitable word:
Ethene and ethyne are examples of ..... hydrocarbons.
What is the next higher homologue of methanol (CH3OH)?
Give the structure of the second member of the alcohol group.
The general formula of Alkane is _________________
While going in an increasing order there is a rise in the molecular mass of the consecutive members of the homologous series by _______.
The general molecular formula for the homologous series of alkynes is _______.
Saturated hydrocarbon : Single bond : : Unsaturated hydrocarbon : _______
Observe the structural formula and answer the following questions.
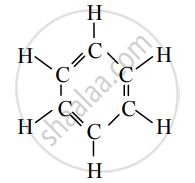
- Write the name of the given hydrocarbon.
- The given hydrocarbon is included in which type of hydrocarbon?
- What is the kind of compounds with the above characteristic structure called?
