Advertisements
Advertisements
प्रश्न
An organic compound having the molecular formula C3H6O can exist in the form of two isomers A and B having different functional groups. The isomer A is a liquid which is used as a solvent for nail polish. The isomer B is also a liquid. An aqueous solution of one of the lower homologues of B is used for preserving biological specimens in the laboratory
(a) What is compound A?
(b) Write the electron-dot structure of A.
(c) What is compound B?
(d) Write the electron-dot structure of B.
(e) Name the lower homologue of compound B which is used in preserving biological specimens.
Advertisements
उत्तर
(a) The isomer A is propanone (acetone).
(b) The electron-dot structure of propanone is::
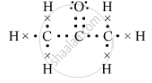
(c) The isomer B is propanal.
(d)The electron-dot structure of propanal is:
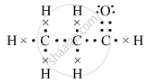
(e) Methanal or formaldehyde is the lower homologue of compound B, which is used to preserve biological specimens.
APPEARS IN
संबंधित प्रश्न
Write the name and formula of the 2nd member of homologous series having general formula CnH2n.
Write the name and formula of the 2nd member of homologous series having general formula CnH2n + 2.
Write the molecular formula of first two members of homologous series having functional group -Cl.
Write the molecular formula of the 2nd and 3rd member of the homologous series where the first member is ethyne.
What is the next higher homologue of methanol (CH3OH)?
Write the names and formulae for the first three members of the homologous series for chloroalkanes.
Define a homologous series. Give the name and structural formula of one homologue of the following:
CH3OH
What is a homologous series? Explain with an example.
Give the dot diagram of the first member of the alcohol.
What is a homologous series?
Haloalkanes react with alkalies to produce alcohol. Give the equation for the preparation of the second member of the homologous series of alcohol. State under what condition the reaction occurs.
The phenomenon in which compounds having different structural formulae have the same molecular formula is called _______.
The general molecular formula for the homologous series of alkynes is _______.
Find the odd one out and give its explanation.
As one ascends in any homologous series, physical properties change gradually.
Which of the following pairs can be the successive members of a homologous series?
Which of the following does not belong to the same homologous series?
Consider the carbon compounds having following molecular formula:
(i) C3H6 (ii) C3H8 (iii) C4H6 (iv) C6H6 (v) C6H12
- State the number of double covalent bonds present in C3H6.
- Write the formula of first member of the homologous series to which the carbon compound C4H6 belongs.
- Which one of the above compounds forms a ring structure of carbon atoms?
- Identify, which of the above compounds, is a member of alkane series.
Name the third homologue of aldehydes.
