Advertisements
Advertisements
प्रश्न
Consider the following molecular formulae of carbon compounds:
(i) CH3COOH (ii) CH3OH (iii) C2H6 (iv) C3H4 (v) C4H8
- Which one of these compounds belongs to homologous series of alcohols?
- Identify the compound having triple bond between carbon-carbon atoms.
- Write the molecular formula of the first member of the homologous series to which CH3COOH belongs.
- Write the general formula of the series to which the compound C4H8 belongs.
Advertisements
उत्तर
- Methanol (CH3OH) belongs to homologous series of alcohols.
- C3H4 has triple bond between carbon atoms.
- Methanoic acid (HCOOH)
- General formula of C4H8 is CnH2n.
APPEARS IN
संबंधित प्रश्न
What is meant by homologous series of carbon compounds?
Write the name and formula of the 2nd member of homologous series having general formula CnH2n – 2.
Write the molecular formula of first two members of homologous series having functional group −Br.
What is the next higher homologue of methanol (CH3OH)?
Write the names and formulae for the first three members of the homologous series for chloroalkanes.
Why homologous series of carbon compounds are so called? Write chemical formula of two consecutive members of a homologous series and state the part of these compounds that determines their (i) physical properties, and (ii) chemical properties.
Haloalkanes react with alkalies to produce alcohol. Give the equation for the preparation of the second member of the homologous series of alcohol. State under what condition the reaction occurs.
Observe the structural formula and answer the following questions.
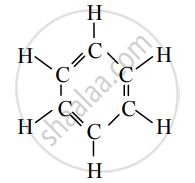
- Write the name of the given hydrocarbon.
- The given hydrocarbon is included in which type of hydrocarbon?
- What is the kind of compounds with the above characteristic structure called?
Consider the carbon compounds having following molecular formula:
(i) C2H2 (ii) C2H5 (iii) C3H7OH (iv) C2H6COOH (v) CH3CHO
- Identify which one of the above compounds, is a member of aldehyde series.
- Write the general formula of the series to which compound C2H2 belongs.
- Which one of the above compounds has triple bonds between carbon-carbon atoms?
- Write the molecular formula of the first member of the homologous series to which the compound C3H7OH belongs.
