Topics
Force, Work, Power and Energy
Force
Light
Work, Energy and Power
- Concept of Work
- Measurement of Work
- Expression for Work (W = F S cosθ)
- Work Done by the Force of Gravity (W = mgh)
- Concept of Power
- Work vs Power
- Concept of Energy
- Energy vs Power
- Mechanical Energy > Potential Energy (U)
- Mechanical Energy > Kinetic Energy (K)
- Potential vs Kinetic Energy
- Conversion of Potential Energy into Kinetic Energy
- Forms of Energy > Solar Energy
- Forms of Energy > Heat Energy
- Forms of Energy > Light Energy
- Forms of Energy > Chemical Energy
- Forms of Energy > Hydro Energy
- Forms of Energy > Electrical Energy
- Forms of Energy > Nuclear Energy
- Forms of Energy > Geo Thermal Energy
- Forms of Energy > Wind Energy
- Forms of Energy > Sound Energy
- Forms of Energy > Magnetic Energy
- Forms of Energy > Mechanical Energy
- Conversion of Energies
- Principle of Conservation of Energy
- Proof: Kinetic + Potential Energy = Constant for Free Fall
- Application of the Principle of Conservation of Energy
Machines
- Concept of Machines
- Technical Terms Related to a Machine
- Principle of a Machine
- Efficiency, Mechanical Advantage, and Velocity Ratio
- Levers
- Types of Levers
- Pulley
- Single Fixed Pulley
- A Single Movable Pulley
- Single Pulley vs Single Movable Pulley
- Combination of Pulleys
- Using one fixed pulley and other movable pulleys
- Using several pulleys in two blocks (block and tackle system)
Sound
Refraction of Light at Plane Surfaces
- Light: Reflection and Refraction
- Refraction of Light
- Laws of Refraction
- Speed of Light in Different Media
- Principle of Reversibility of the Path of Light
- Refraction Laws & Glass Index
- Refraction of Light Through a Rectangular Glass Block
- Multiple Images in a Thick Mirror
- Prism
- Refraction of Light Through a Prism
- Real and Apparent Depth
- Apparent Bending of a Stick Under Water
- Consequences of Refraction of Light
- Transmission of Light From a Denser Medium to a Rarer Medium
- Critical Angle
- Relationship between Critical Angle and Refractive Index
- Total Internal Reflection
- Total Internal Reflection in a Prism
- Total Internal Reflection Through a Right-Angled Isosceles Prism
- Total Internal Reflection Through an Equilateral Prism
- Total Internal Reflection Through Right-angled prism
- Use of a Total Internal Reflecting Prism in Place of a Plane Mirror
- Total Internal Reflection vs Reflecting from a Plane Mirror
- Consequences of Total Internal Refraction
Electricity and Magnetism
Refraction Through a Lens
- Concept of Lenses
- Action of a Lens as a Set of Prisms
- Technical Terms Related to a Lens
- Convex Lens vs Concave Lens
- Refraction of Light Through an Equi-Convex Lens and an Equi-Concave Lens
- Principal Rays for Ray Diagrams
- Real Image vs Virtual Image
- Construction of a Ray Diagram for a Lens
- Images Formed by Convex Lenses
- Images Formed by Concave Lenses
- Sign Convention
- Lens Formula
- Linear Magnification
- Power of a Lens
- Magnifying Glass Or Simple Microscope
- Application of Lenses
- Experimental Determination of Focal Length of Convex Lens
- Convex Lens vs Concave Lens
Heat
Modern Physics
Spectrum
- Deviation Produced by a Triangular Prism
- Colour in White Light with Their Wavelength and Frequency Range
- Dispersion of Light
- Electromagnetic Spectrum
- Properties and Uses of Different Radiations of the Electromagnetic Spectrum
- Distinction between Ultraviolet, Visible, and Infrared Radiations
- Scattering of Light
- Applications of Scattering of Light
Sound
- Sound Waves
- Light Waves vs Sound Waves
- Reflection of Sound Waves
- Echo
- Determination of Speed of Sound by the Method of Echo
- Use of Echoes
- Natural Vibrations
- Damped Vibrations
- Natural Vibrations vs Damped Vibrations
- Forced Vibrations
- Natural Vibrations vs Forced Vibrations
- Resonance (a special case of forced vibrations)
- Demonstration of Resonance
- Forced Vibrations vs Resonant Vibrations
- Examples of Resonance
- Characteristics of Sound
- Loudness and Intensity
- Pitch and frequency
- Quality and Wave Form
- Music and Noise
Current Electricity
- Electric Charge
- Electric Current
- Potential and Potential Difference
- Electric Resistance
- Ohm's Law
- Experimental Verification of Ohm’s Law and Ohmic Resistors
- Ohmic and Non-ohmic Resistors
- Specific Resistance
- Choice of Material of a Wire
- Superconductors
- Electro-Motive Force of a Cell
- Terminal Voltage of a Cell
- Internal Resistance of a Cell
- Resistance of a System of Resistors
- Resistors in Series
- Resistors in Parallel
- A combination of resistors in both series and parallel
- Forms of Energy > Electrical Energy
- Measurement of Electrical Energy
- Electrical Power
- Commercial Unit of Electrical Energy
- Power Rating of Common Electrical Appliances
- Household Consumption of Electric Energy
- Heating Effect of Electric Current
Household Circuits
- Transmission of Power from the Power Generating Station to the Consumer
- Power Distribution to a House
- House Wiring (Ring System)
- Fuse
- Reason for connecting the fuse in the live wire
- Current Rating of a Fuse
- Switches
- Circuits with Dual Control Switches (Staircase Wire)
- Earthing
- Three-pin Plug and Socket
- Colour Coding of Wires in a Cable
- High Tension Wires
- Precautions to Be Taken While Using Electricity
Electro-Magnetism
- Oersted's Experiment
- Applications of Biot-Savart's Law > Magnetic Field due to a Finite Straight Current-Carrying Wire
- Right-hand Thumb Rule
- Applications of Biot-Savart's Law > Magnetic Field at the Centre of a Circular Loop
- Applications of Ampere’s Circuital Law > Magnetic Field of a Long Straight Solenoid
- Electromagnet
- Permanent Magnet
- Comparison of an Electro Magnet with a Permanent Magnet
- Advantages of an Electromagnet over a Permanent Magnet
- Uses of Electromagnet
- Force on a Current Carrying Conductor in a Magnetic Field
- Simple D.C. Motor
- Electromagnetic Induction
- Demonstration of the Phenomenon of Electromagnetic Induction
- Faraday's Explanation
- Faraday's Laws of Electromagnetic Induction
- A.C. Generator
- Frequency of an a.c. in Household Supplies
- Comparison Between A.C. Generator and D.C. Motor
- Transformers
Calorimetry
- Heat
- The Temperature and a Thermometer
- Factors Affecting the Quantity of Heat Absorbed to Increase the Temperature of a Body
- Heat vs Temperature
- Thermal or Heat Capacity
- Specific Heat Capacity
- Heat Capacity vs Specific Heat Capacity
- Specific Heat Capacity of Some Common Substances
- Calorimetry
- Principle of Method of Mixtures or Principle of Calorimetry
- Natural Phenomena and Consequences of High Specific Heat Capacity of Water
- Examples of High and Low Heat Capacity
- Change of State
- Melting and Freezing
- Melting Point and Its Effects
- Vaporisation or Boiling
- Boiling Point and Its Effects
- Latent Heat
- Specific Latent Heat of Fusion of Ice
- Explanation of Latent Heat of Melting based on Kinetic Model
- Natural Consequences of High Specific Latent Heat of Fusion of Ice
Radioactivity
- Structure of the Atom and Nucleus
- Atomic Models
- Isotopes
- Isobars
- Isotones
- Radioactivity
- Radioactivity as emission of Alpha, Beta, and Gamma Radiations
- Properties of Alpha Particles
- Properties of Beta Particles
- Properties of Gamma Radiations
- Distinction between the Properties of α, β, and γ Radiations
- Changes Within the Nucleus in Alpha, Beta and Gamma Emission
- Uses of Radioactive Isotopes
- Sources of Harmful Radiations
- Harmful Effects of Radiation
- Safety Precautions While Using Nuclear Energy
- Background Radiations
- Forms of Energy > Nuclear Energy
- Nuclear Fission
- Radioactive Decay Vs Nuclear Fission
- Nuclear Fusion
- Nuclear Fission Vs Nuclear Fusion
- Introduction
- Definition: Change of State
- The Six Types of Phase Changes
- Practical Applications
- Key Points: Practical Applications of State
Introduction
Matter exists in three common states: solid, liquid, and gas. A transition from one state to another is called a change of state (or phase change). These transitions are driven by heat exchange between a substance and its surroundings.
When heat is added to a substance, its molecules gain kinetic energy and vibrate more vigorously. At certain critical temperatures, the supplied energy goes into breaking intermolecular bonds rather than raising the temperature. This is why temperature pauses during a phase change, even though heat continues to flow in.
CISCE: Class 10
Definition: Change of State
The process of change from one state to another at a constant temperature is called the change of phase.
OR
A transition from one state of matter (solid, liquid, or gas) to another is called change of state.
The Six Types of Phase Changes
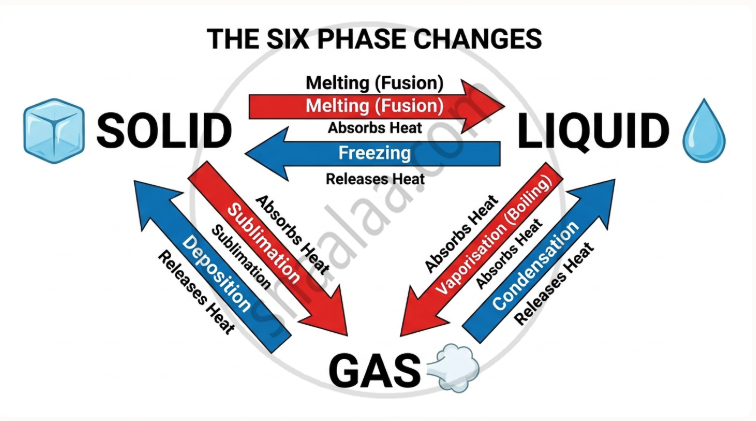
 All possible transitions between solid, liquid, and gas — and their reverse — are summarised below.
All possible transitions between solid, liquid, and gas — and their reverse — are summarised below.
| Phase Change | Direction | Process Name | Energy | Example |
|---|---|---|---|---|
| Solid → Liquid | Gains heat | Melting (Fusion) | Endothermic | Ice → Water at 0 °C |
| Liquid → Solid | Loses heat | Freezing | Exothermic | Water → Ice at 0 °C |
| Liquid → Gas | Gains heat | Vaporisation (Boiling) | Endothermic | Water → Steam at 100 °C |
| Gas → Liquid | Loses heat | Condensation | Exothermic | Steam → Water droplets |
| Solid → Gas | Gains heat | Sublimation | Endothermic | Dry ice (CO₂), Naphthalene mothballs |
| Gas → Solid | Loses heat | Deposition | Exothermic | Frost forming on cold surfaces |
Practical Applications
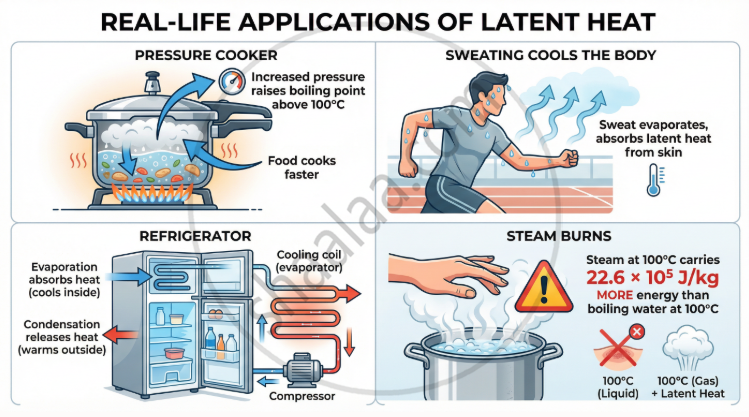
- Pressure cooker: Increased pressure raises the boiling point above 100 °C, so food cooks faster at higher temperature.
- Cooking at hill stations: Lower atmospheric pressure means water boils 100 °C below, making cooking slower.
- Vacuum distillation: Reducing pressure lowers boiling points, allowing heat-sensitive substances to be distilled without decomposition.
Key Points: Practical Applications of State
- A change of state occurs when heat exchange causes a substance to transition between solid, liquid, and gas phases.
- Temperature remains constant during a phase change because heat energy changes molecular arrangement (potential energy), not molecular speed (kinetic energy).
- The heating curve has flat plateaus at the melting point (0 °C) and boiling point (100 °C) for water, with rising slopes in between.
