Advertisements
Advertisements
Complete the following reactions:

Concept: Chemical Reactions of Aldehydes and Ketones - Other Reactions
Name the reagents used in the following reactions:

Concept: Methods of Preparation of Carboxylic Acids
Why are alkyl halides insoluble in water?
Concept: Physical Properties of Carboxylic Acids
How do you convert the following :
Ethyne to Ethanal
Concept: Nomenclature of Carboxylic Acids
Write the equations involved in the following reactions :
Stephen reaction
Concept: Nomenclature of Carboxylic Acids
Predict the products of the following reactions :
Concept: Chemical Reactions of Aldehydes and Ketones - Reduction
Concept: Chemical Reactions of Aldehydes and Ketones - Oxidation
Predict the products of the following reactions :
Concept: Chemical Reactions of Aldehydes and Ketones - Reduction
Write the IUPAC name of the follow
\[\begin{array}\ce{\phantom{--..}O}\\\ce{\phantom{--..}||}\\ \ce{CH2=CH-C-CH3}\end{array}\]
Concept: Preparation of Aldehydes and Ketones
What happens when Salicylic acid is treated with (CH3CO)2 O/H+?
Concept: Chemical Reactions of Carboxylic Acids - Reactions Involving Cleavege of O-H Bond
Predict the main product of the following reaction:\[\begin{array}{c}
\ce{O\phantom{----}O\phantom{-}}\\
\ce{||\phantom{----}||\phantom{-}}\\
\ce{CH3-C-CH2-C-OCH3}
\end{array}\ce{->[(i)NaBH4][(ii)H+]}\]
Concept: Chemical Reactions of Aldehydes and Ketones - Reduction
Why is alpha (α) hydrogen of carbonyl compounds acidic in nature?
Concept: Chemical Reactions of Aldehydes and Ketones - Reactions Due to α-hydrogen
Give a simple chemical test to distinguish between 
Concept: Chemical Reactions of Aldehydes and Ketones - Nucleophilic Addition Reactions
Write the main product formed when propanal reacts with the following reagents:
2 moles of 3 CH OH in presence of dry HCl
Concept: Chemical Reactions of Aldehydes and Ketones - Nucleophilic Addition Reactions
Write the main product formed when propanal reacts with the following reagents:
H2N- NH2 followed by heating with KOH in ethylene glycol.
Concept: Chemical Reactions of Aldehydes and Ketones - Nucleophilic Addition Reactions
Arrange the following compounds in increasing order of their property as indicated:
F - CH2COOH, O2N - CH2 COOH CH3 COOH,HCOOH - acid character.
Concept: Chemical Reactions of Carboxylic Acids - Reactions Involving Cleavege of O-H Bond
Acetone, Acetaldehyde, Benzaldehyde, Acetophenone – reactivity towards addition of HCN.
Concept: Chemical Reactions of Aldehydes and Ketones - Nucleophilic Addition Reactions
complete the following reaction:
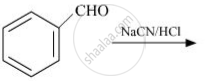
Concept: Chemical Reactions of Aldehydes and Ketones - Other Reactions
complete the following reaction:
\[\begin{array}{cc}
\phantom{...}\ce{CH3} \\
| \\
\phantom{.................}\ce{CH3-CH-COOH ->[(i) Br2/Red P4][(ii)H2O]}
\end{array}\]
Concept: Chemical Reactions of Aldehydes and Ketones - Other Reactions
Give a reason for the following :
N-N bond is weaker than the P-P bond.
Concept: Chemical Reactions of Carboxylic Acids - Reactions Involving –COOH Group
