Please select a subject first
Advertisements
Advertisements
A 12.9 eV beam of electronic is used to bombard gaseous hydrogen at room temperature. Upto which energy level the hydrogen atoms would be excited ?
Calculate the wavelength of the first member of Paschen series and first member of Balmer series.
Concept: Energy Levels
A 12.3 eV electron beam is used to bombard gaseous hydrogen at room temperature. Upto which energy level the hydrogen atoms would be excited?
Calculate the wavelengths of the second member of Lyman series and second member of Balmer series.
Concept: Energy Levels
An electron in an atom revolves round the nucleus in an orbit of radius r with frequency v. Write the expression for the magnetic moment of the electron.
Concept: Alpha-particle Scattering and Rutherford’s Nuclear Model of Atom
Using Bohr’s postulates, obtain the expressions for (i) kinetic energy and (ii) potential energy of the electron in stationary state of hydrogen atom.
Draw the energy level diagram showing how the transitions between energy levels result in the appearance of Lymann Series.
Concept: Bohr’s Model for Hydrogen Atom
Using Bohr’s postulates, obtain the expression for the total energy of the electron in the stationary states of the hydrogen atom. Hence draw the energy level diagram showing how the line spectra corresponding to Balmer series occur due to transition between energy levels.
Concept: Bohr’s Model for Hydrogen Atom
Using Bohr’s postulates, obtain the expression for total energy of the electron in the nth orbit of hydrogen atom.
Concept: Bohr’s Model for Hydrogen Atom
Draw the energy level diagram showing how the line spectra corresponding to Paschen series occur due to transition between energy levels.
Concept: Energy Levels
In a Geiger-Marsden experiment, calculate the distance of closest approach to the nucleus of Z = 80, when a α-particle of 8Mev energy impinges on it before it comes momentarily to rest and reverses its direction.
How will the distance of closest approach be affected when the kinetic energy of the α-particle is doubles?
Concept: Alpha-particle Scattering and Rutherford’s Nuclear Model of Atom
The ground state energy of hydrogen atom is −13.6 eV. If and electron make a transition from the energy level −0.85 eV to −3.4 eV, calculate spectrum does his wavelength belong?
Concept: Hydrogen Spectrum
In a Geiger-Marsden experiment, calculate the distance of closest approach to the nucleus of Z = 75, when a α-particle of 5 MeV energy impinges on it before it comes momentarily to rest and reverses its direction.
How will the distance of closest approach be affected when the kinetic energy of the α-particle is doubles?
Concept: Alpha-particle Scattering and Rutherford’s Nuclear Model of Atom
The ground state energy of hydrogen atom is −13.6 eV. If an electron make a transition from an energy level −0.85 eV to −1.51 eV, calculate the wavelength of the spectral line emitted. To which series of hydrogen spectrum does this wavelength belong?
Concept: Hydrogen Spectrum
Write the expression for Bohr’s radius in hydrogen atom ?
Concept: Bohr’s Model for Hydrogen Atom
The energy levels of an atom are as shown below. Which of them will result in the transition of a photon of wavelength 275 nm?
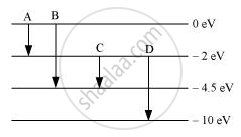
Concept: Energy Levels
Which transition corresponds to emission of radiation of maximum wavelength?
Concept: Energy Levels
Plot a graph showing the variation of de Broglie wavelength (X) associated with a charged a particle of mass m, versus `1/sqrtV` where V is the potential difference through which the particle is accelerated. How does this graph give us information regarding the magnitude of the charge of the particle?
Concept: De Broglie’s Explanation of Bohr’s Second Postulate of Quantisation
Answer the following question.
A charged particle q is moving in the presence of a magnetic field B which is inclined to an angle 30° with the direction of the motion of the particle. Draw the trajectory followed by the particle in the presence of the field and explain how the particle describes this path.
Concept: Alpha-particle Scattering and Rutherford’s Nuclear Model of Atom
Answer the following question.
Explain briefly how Rutherford scattering of α-particle by a target nucleus can provide information on the size of the nucleus.
Concept: Alpha-particle Scattering and Rutherford’s Nuclear Model of Atom
When the electron orbiting in hydrogen atom in its ground state moves to the third excited state, show how the de Broglie wavelength associated with it would be affected.
Concept: Bohr’s Model for Hydrogen Atom
Consider two different hydrogen atoms. The electron in each atom is in an excited state. Is it possible for the electrons to have different energies but same orbital angular momentum according to the Bohr model? Justify your answer.
Concept: Bohr’s Model for Hydrogen Atom
Derive an expression for the frequency of radiation emitted when a hydrogen atom de-excites from level n to level (n – 1). Also show that for large values of n, this frequency equals to classical frequency of revolution of an electron.
Concept: Bohr’s Model for Hydrogen Atom
