Advertisements
Advertisements
Question
Using Bohr’s postulates, obtain the expressions for (i) kinetic energy and (ii) potential energy of the electron in stationary state of hydrogen atom.
Draw the energy level diagram showing how the transitions between energy levels result in the appearance of Lymann Series.
Advertisements
Solution
According to Bohr’s postulates, in a hydrogen atom, a single alectron revolves around a nucleus of charge +e. For an electron moving with a uniform speed in a circular orbit os a given radius, the centripetal force is provided by Columb force of attraction between the electron and the nucleus. The gravitational attraction may be neglected as the mass of electron and proton is very small.
So,
`mv^2/r = (ke^2)/r^2`
0r `mv^2 = (ke^2)/r ...................(1)`
where m = mass of electron
r = radius of electronic orbit
v = velocity of electron.
Again,
`mvr = (nh)/(2π)`
` or v = (nh)/(2πmr)`
From eq(1), we get,
`m ((nh)/(2πmr))^2 = (ke^2)/r`
`⇒ r = (n^2h^2)/(4π^2kme^2) .....................(2) `
(i) Kinetic energy of electron,
`E_k = 1/2mv^2 = (ke^2)/(2r)`
Using eq (2), we get
`E_k = (ke^2)/2 (4π^2kme^2)/(n^2h^2)`
= `(2π^2k^2me^4)/(n^2h^2)`
(ii) Potential energy
`E^p= - ke^2 xx (4π^2k^2me^4)/(n^2h^2) `
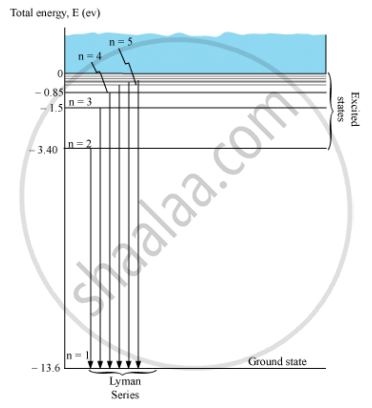
Energy level diagram showing the transitions between energy levels result in the appearance ofLymann series:
For Lymann series, nf = 1 and ni = 2, 3, 4, 5, …
`1/λ = R_H(1/I^2 - 1/n^2)`
Where, ni = 2, 3, 4, 5, …
APPEARS IN
RELATED QUESTIONS
The radius of the innermost electron orbit of a hydrogen atom is 5.3 × 10−11 m. What are the radii of the n = 2 and n = 3 orbits?
What is the energy in joules released when an electron moves from n = 2 to n = 1 level in a hydrogen atom?
Derive an expression for the frequency of radiation emitted when a hydrogen atom de-excites from level n to level (n – 1). Also show that for large values of n, this frequency equals to classical frequency of revolution of an electron.
In form of Rydberg's constant R, the wave no of this first Ballmer line is
The angular momentum of electron in nth orbit is given by
An electron in H-atom makes a transition from n = 3 to n = 1. The recoil momentum of the H-atom will be ______.
The energy required to remove the electron from a singly ionized Helium atom is 2.2 times the energy required to remove an electron from Helium atom. The total energy required to ionize the Helium atom completely is ______.
In hydrogen atom, transition from the state n = 6 to n = 1 results in ultraviolet radiation. Infrared radiation will be obtained in the transition ______.
State three postulates of Bohr's theory of hydrogen atom.
Find the angular momentum of an electron revolving in the second orbit in Bohr's hydrogen atom.
