Advertisements
Advertisements
प्रश्न
Using Bohr’s postulates, obtain the expressions for (i) kinetic energy and (ii) potential energy of the electron in stationary state of hydrogen atom.
Draw the energy level diagram showing how the transitions between energy levels result in the appearance of Lymann Series.
Advertisements
उत्तर
According to Bohr’s postulates, in a hydrogen atom, a single alectron revolves around a nucleus of charge +e. For an electron moving with a uniform speed in a circular orbit os a given radius, the centripetal force is provided by Columb force of attraction between the electron and the nucleus. The gravitational attraction may be neglected as the mass of electron and proton is very small.
So,
`mv^2/r = (ke^2)/r^2`
0r `mv^2 = (ke^2)/r ...................(1)`
where m = mass of electron
r = radius of electronic orbit
v = velocity of electron.
Again,
`mvr = (nh)/(2π)`
` or v = (nh)/(2πmr)`
From eq(1), we get,
`m ((nh)/(2πmr))^2 = (ke^2)/r`
`⇒ r = (n^2h^2)/(4π^2kme^2) .....................(2) `
(i) Kinetic energy of electron,
`E_k = 1/2mv^2 = (ke^2)/(2r)`
Using eq (2), we get
`E_k = (ke^2)/2 (4π^2kme^2)/(n^2h^2)`
= `(2π^2k^2me^4)/(n^2h^2)`
(ii) Potential energy
`E^p= - ke^2 xx (4π^2k^2me^4)/(n^2h^2) `
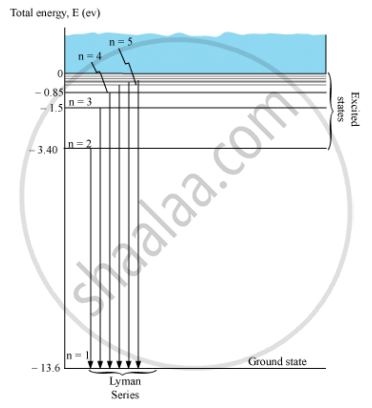
Energy level diagram showing the transitions between energy levels result in the appearance ofLymann series:
For Lymann series, nf = 1 and ni = 2, 3, 4, 5, …
`1/λ = R_H(1/I^2 - 1/n^2)`
Where, ni = 2, 3, 4, 5, …
APPEARS IN
संबंधित प्रश्न
A positive ion having just one electron ejects it if a photon of wavelength 228 Å or less is absorbed by it. Identify the ion.
A parallel beam of light of wavelength 100 nm passes through a sample of atomic hydrogen gas in ground state. (a) Assume that when a photon supplies some of its energy to a hydrogen atom, the rest of the energy appears as another photon. Neglecting the light emitted by the excited hydrogen atoms in the direction of the incident beam, what wavelengths may be observed in the transmitted beam? (b) A radiation detector is placed near the gas to detect radiation coming perpendicular to the incident beam. Find the wavelengths of radiation that may be detected by the detector.
Suppose in an imaginary world the angular momentum is quantized to be even integral multiples of h/2π. What is the longest possible wavelength emitted by hydrogen atoms in visible range in such a world according to Bohr's model?
What is the energy in joules released when an electron moves from n = 2 to n = 1 level in a hydrogen atom?
The energy associated with the first orbit of He+ is ____________ J.
Why was a change in the Bohr Model of atom required? Due to which important development (s), concept of movement of an electron in an orbit was replaced by, the concept of probability of finding electron in an orbital? What is the name given to the changed model of atom?
The electron in a hydrogen atom first jumps from the third excited state to the second excited state and subsequently to the first excited state. The ratio of the respective wavelengths, λ1/λ2, of the photons emitted in this process is ______.
The line at 434 nm in the Balmer series of the hydrogen spectrum corresponds to a transition of an electron from the nth to second Bohr orbit. The value of n is ______.
According to Bohr's theory, the radius of the nth Bohr orbit of a hydrogen like atom of atomic number Z is proportional to ______.
The de Broglie wavelength of an electron in the first Bohr’s orbit of hydrogen atom is equal to ______.
