Advertisements
Advertisements
Question
Answer the following question.
A charged particle q is moving in the presence of a magnetic field B which is inclined to an angle 30° with the direction of the motion of the particle. Draw the trajectory followed by the particle in the presence of the field and explain how the particle describes this path.
Advertisements
Solution

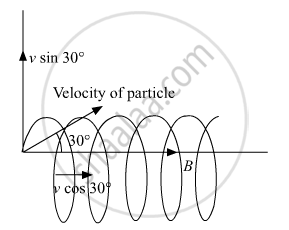
Lorentz force will be exerted on the particle because of the Sine component of particle velocity thereby making the particle move in a helical path.
Time period = `("Total distance")/("speed") = (2"πR")/("v"_"y") (2"πR")/("v" sin 30°) = (4"πR")/("v")`
Pitch = speed along x-axis × time period
`= "v"_x xx (2"πR")/("v"_"y")`
`= "v"_x xx (2"πR")/("v"sin30°) = "v"_x xx (2"πR" xx 2)/("v")`
= `"v" cos 30° xx (4"πR")/("v") = sqrt(3)/(2) xx 4"πR" = 2sqrt(3)"πR"`
R = `(mv sin 30°)/(q"B")`
Pitch = `(2sqrt(3)"πmv" sin 30°)/(q"B")`
= `(2sqrt(3)"πmv")/(q"B" xx2)`
= `(sqrt(3)"πmv")/(q"B")`
The vertical component of velocity will make the charged particle to move in a circular path whereas the horizontal component of velocity will provide Pitch = `(sqrt(3)"πmv")/(q"B")` Hence the motion of the particle will be helical with the Pitch= `(sqrt(3)"πmv")/(q"B")`.
APPEARS IN
RELATED QUESTIONS
Thorium 90Th232 is disintegrated into lead 82Pb200. Find the number of α and β particles emitted in disintegration.
Answer the following question, which help you understand the difference between Thomson’s model and Rutherford’s model better.
Is the probability of backward scattering (i.e., scattering of α-particles at angles greater than 90°) predicted by Thomson’s model much less, about the same, or much greater than that predicted by Rutherford’s model?
Alpha particles used in Geiger-Marsden experiment were obtained from ______.
The basic force acting on the alpha particles using Coulomb's law is ______.
Which one of the following statements does not hold good when two balls of masses m1 and m2 undergo elastic collision?
The ratio active Nude 7N13 decays 6C13 through the emission of
Would the Bohr formula for the H-atom remain unchanged if proton had a charge (+4/3)e and electron a charge (−3/4)e, where e = 1.6 × 10–19C. Give reasons for your answer.
Draw a graph showing the variation of the number of particles scattered (N) with the scattering angle θ in the Geiger-Marsden experiment. Why only a small fraction of the particles are scattered at θ > 90°?
Choose the correct option from the following options given below:
Determine the distance of the closest approach when an alpha particle of kinetic energy 3.95 MeV approaches a nucleus of Z = 79, stops and reverses its directions.
