Advertisements
Advertisements
प्रश्न
Answer the following question.
A charged particle q is moving in the presence of a magnetic field B which is inclined to an angle 30° with the direction of the motion of the particle. Draw the trajectory followed by the particle in the presence of the field and explain how the particle describes this path.
Advertisements
उत्तर

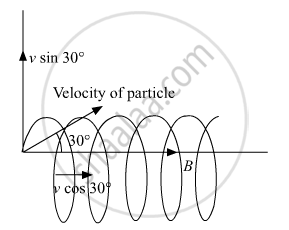
Lorentz force will be exerted on the particle because of the Sine component of particle velocity thereby making the particle move in a helical path.
Time period = `("Total distance")/("speed") = (2"πR")/("v"_"y") (2"πR")/("v" sin 30°) = (4"πR")/("v")`
Pitch = speed along x-axis × time period
`= "v"_x xx (2"πR")/("v"_"y")`
`= "v"_x xx (2"πR")/("v"sin30°) = "v"_x xx (2"πR" xx 2)/("v")`
= `"v" cos 30° xx (4"πR")/("v") = sqrt(3)/(2) xx 4"πR" = 2sqrt(3)"πR"`
R = `(mv sin 30°)/(q"B")`
Pitch = `(2sqrt(3)"πmv" sin 30°)/(q"B")`
= `(2sqrt(3)"πmv")/(q"B" xx2)`
= `(sqrt(3)"πmv")/(q"B")`
The vertical component of velocity will make the charged particle to move in a circular path whereas the horizontal component of velocity will provide Pitch = `(sqrt(3)"πmv")/(q"B")` Hence the motion of the particle will be helical with the Pitch= `(sqrt(3)"πmv")/(q"B")`.
APPEARS IN
संबंधित प्रश्न
Using Rutherford's model of the atom, derive the expression for the total energy of the electron in hydrogen atom. What is the significance of total negative energy possessed by the electron?
In a Geiger-Marsden experiment, calculate the distance of closest approach to the nucleus of Z = 75, when a α-particle of 5 MeV energy impinges on it before it comes momentarily to rest and reverses its direction.
How will the distance of closest approach be affected when the kinetic energy of the α-particle is doubles?
The total energy of an electron in the ground state of the hydrogen atom is -13·6 eV. Its total energy, when a hydrogen atom is in the first excited state, is ______.
In Geiger-Marsden experiment prediction was that ______.
The model that best explains the results of Geiger-Marsden experiment is ______.
Plutonium decays with half of 24000 years. If plutonium is store for 72000 yrs. The fraction of .its that remain:-
O2 molecule consists of two oxygen atoms. In the molecule, nuclear force between the nuclei of the two atoms ______.
Draw a graph showing the variation of the number of particles scattered (N) with the scattering angle θ in the Geiger-Marsden experiment. Why only a small fraction of the particles are scattered at θ > 90°?
An alpha nucleus of energy `1/2`mv2 bombards a heavy nuclear target of charge Ze. Then the distance of closest approach for the alpha nucleus will be proportional to ______.
- v2
- `1/"m"`
- `1/"v"^2`
- `1/"Ze"`
Determine the distance of the closest approach when an alpha particle of kinetic energy 3.95 MeV approaches a nucleus of Z = 79, stops and reverses its directions.
