Advertisements
Advertisements
F2 has lower bond dissociation enthalpy than Cl2. Why?
Concept: Group 17 Elements - The Halogen Family
Which noble gas is used in filling balloons for meteorological observations?
Concept: Group 18 Elements - The Noble gas Family
Complete the equation : XeF2+PF5 →
Concept: Group 18 Elements - The Noble gas Family
Give reasons for the following : Oxygen has less electron gain enthalpy with negative sign than sulphur.
Concept: Group 16 Elements - The Oxygen Family
Give reasons for the following : H3PO2 is a stronger reducing agent than H3PO3.
Concept: Group 15 Elements - The Nitrogen Family >> Oxoacids of Phosphorus
Which halogen compound in the following pair will react faster in SN2 reaction CH3Br or CH3I.
Concept: Group 17 Elements - The Halogen Family
Which halogen compound in each of the following pairs will react faster in SN2 reaction
(CH3)3 C – Cl or CH3 – Cl
Concept: Group 17 Elements - The Halogen Family
Draw the structure of the following:
H2SO4
Concept: Group 16 Elements - The Oxygen Family >> Sulphuric Acid
What is meant by hydroboration-oxidation reaction? Illustrate it with an example.
Concept: Methods of Preparation of Alcohols
Give reasons Thermal stability decreases from H2O to H2Te.
Concept: Group 16 Elements - The Oxygen Family
Write the product(s) in the following reactions
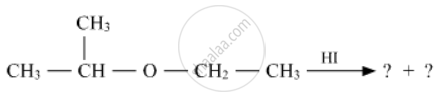
Concept: Physical Properties of Ethers
Write the product(s) in the following reaction
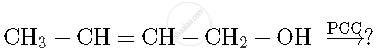
Concept: Chemical Reaction of Ethers - Cleavege of C-O Bonds
Give simple chemical tests to distinguish between the following pairs of compounds :
Propanol and 2-methylpropan-2-ol
Concept: Physical Properties of Ethers
Write the formula of reagents used in the following reactions :
Bromination of phenol to 2,4,6-tribromophenol
Concept: Chemical Reaction of Ethers - Cleavege of C-O Bonds
Write the formula of reagents used in the following reactions :
Hydroboration of propene and then oxidation to propanol.
Concept: Chemical Reaction of Ethers - Cleavege of C-O Bonds
Write the mechanism (using curved arrow notation) of the following reaction :
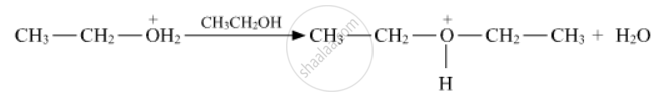
Concept: Chemical Reactions of Alcohols and Phenols >> Reactions Involving Cleavage of Carbon–Oxygen (C–O) Bond in Alcohols
Complete the following chemical equation:
\[\ce{Ca3P2 + H2O ->}\]
Concept: Group 15 Elements - The Nitrogen Family >> Phosphine
How do you prepare:
K2MnO4 from MnO2?
Concept: Group 16 Elements - The Oxygen Family >> Classification of Oxides
How do you prepare:
Na2Cr2O7 from Na2CrO4?
Concept: Group 16 Elements - The Oxygen Family >> Sulphur - Allotropic Forms
What happens when (CH3)3 C – OH is heated with Cu/573 K?
Write the chemical equation in support of your answer.
Concept: Chemical Reactions of Alcohols and Phenols >> Reactions Involving Cleavage of Carbon–Oxygen (C–O) Bond in Alcohols
