Definitions [13]
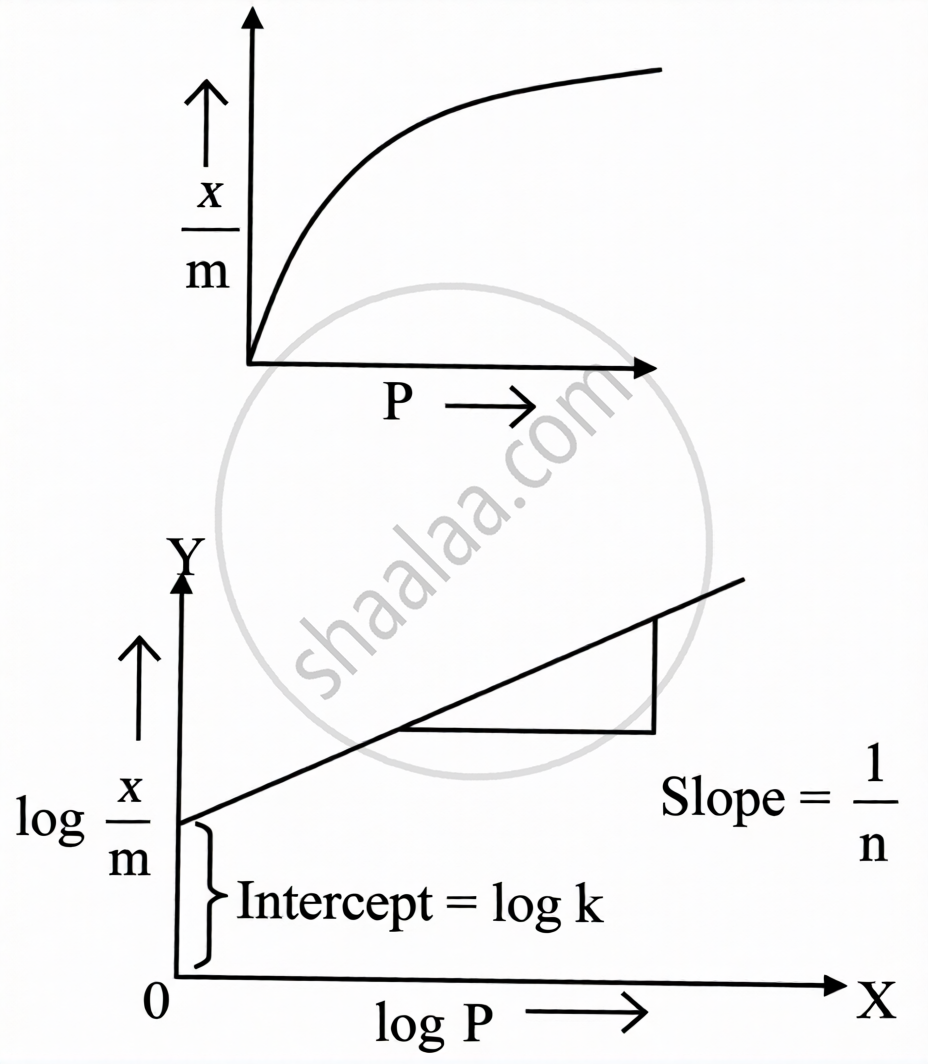
The relationship between the extent of adsorption (x/m) and the pressure of gas at constant temperature is called an adsorption isotherm.
Define the term Electrophoresis.
The movement of colloidal particles under an applied electric potential is called electrophoresis.
A substance is said to be in colloidal state when one substance of heterogeneous system is dispersed (dispersed phase) as very fine particles in another substance (dispersion medium).
Define the following term:
Multimolecular colloids
Multimolecular colloids are species having size in the colloidal range (diameter < 1 nm) that are formed by the aggregation of large number of atoms or small molecules of substances when dissolved in the dispersion medium.
The process used for reducing the amount of impurities to a requisite minimum is called as purification of colloidal solution.
Dialysis process is used for removing a dissolved substance from a colloidal solution by diffusion through a suitable membrane.
The process used for reducing the amount of impurities to a requisite minimum is called as purification of colloidal solution.
Dialysis process is used for removing a dissolved substance from a colloidal solution by diffusion through a suitable membrane.
Define the following term:
Multimolecular colloids
Multimolecular colloids are species having size in the colloidal range (diameter < 1 nm) that are formed by the aggregation of large number of atoms or small molecules of substances when dissolved in the dispersion medium.
Define coagulation.
It is a process of aggregating together the colloidal particles so as to change them into large-sized particles, which ultimately settle as a precipitate.
Define the following term:
Coagulation
Coagulation is the process by which colloidal particles aggregate and settle as a precipitate. It is brought about by the addition of electrolyte.
Define the following term:
Zeta potential
The potential difference between a colloidal particle's fixed layer (primary layer) and diffused layer (secondary layer) is known as the zeta potential. It's also referred to as electrokinetic potential.
Emulsions are colloidal solutions of two immiscible liquids in which one liquid acts as the dispersed phase and the other as the dispersion medium.
Key Points
| Feature | Physisorption | Chemisorption |
|---|---|---|
| Force | Van der Waals' | Covalent / ionic bond |
| Specificity | Non-specific | Highly specific |
| Heat of adsorption | 20–40 kJ mol⁻¹ | 40–200 kJ mol⁻¹ |
| Temperature | Low temp favoured | High temp favoured |
| Reversibility | Reversible | Irreversible |
| Layers | Multilayer | Monolayer |
Freundlich Adsorption Isotherm:
Empirical equation: \[\frac{x}{m}=kP^{1/n}\quad(n>1)\]
where:
- x = mass of the gas adsorbed
- m = mass of the adsorbent
- `x/m` = mass of gas adsorbed per unit mass of adsorbent
- P = equilibrium pressure
- k and n = constants depending on the nature of the adsorbate, adsorbent, and temperature
For solutions: \[\frac{x}{m}=kC^{1/n}\] where C = concentration of the solution
Logarithmic (linear) form: \[\log\frac{x}{m}=\log k+\frac{1}{n}\log P\]
In the log x/m vs log P graph:
- Slope = `1/n` (value between 0 and 1)
- Y-intercept = log k
- When `1/n` = 0: adsorption is independent of pressure
- When `1/n` = 1: adsorption is directly proportional to pressure
| Application | How Adsorption Helps |
|---|---|
| Adsorption indicators | Dyes like eosin and fluorescein act as indicators through adsorption |
| Separation of inert gases | Inert gases can be separated on coconut charcoal (different adsorption at different temperatures) |
| Froth flotation process | Hydrophobic pine oil preferentially adsorbs sulphide ore particles, allowing separation from gangue |
| Chromatographic analysis | Powdered adsorbents like silica or alumina gel are used to separate mixtures |
| Heterogeneous catalysis | Contact process (SO₂ → SO₃), hydrogenation of oils |
| Gas masks | Activated charcoal or mixture of adsorbents to avoid inhaling poisonous gases |
| Control of humidity | Silica and alumina gels used to adsorb moisture |
| Production of high vacuum | Coconut charcoal adsorbs traces of air or moisture to create high vacuum |
The reaction occurs on the surface of the solid catalyst. Steps involved:
- Diffusion — Reactant molecules diffuse towards the surface of the catalyst
- Adsorption — Reactant molecules adsorb onto the surface of the catalyst
- Intermediate formation — Chemical reaction on the catalyst surface proceeds via intermediate formation
- Desorption — Products are desorbed (released) from the catalyst surface
- Product formation — Products diffuse away from the surface
Heterogeneous catalytic reactions are also called surface reactions. Heterogeneous catalysts are used in automobile catalytic converters.
1. Chemical Methods
-
Oxidation: \[\mathrm{SO}_2+2\mathrm{H}_2\mathrm{S}\xrightarrow{\mathrm{Oxidation}}3\mathrm{S}\downarrow+2\mathrm{H}_2\mathrm{O}\] (sulphur sol)
-
Reduction: \[2\mathrm{AuCl}_{3}+3\mathrm{HCHO}+3\mathrm{H}_{2}\mathrm{O}\xrightarrow{\mathrm{Reduction}}2\mathrm{Au}\downarrow+3\mathrm{HCOOH}+6\mathrm{HCl}\] (gold sol)
- Hydrolysis: \[\mathrm{FeCl}_3+3\mathrm{H}_2\mathrm{O}\xrightarrow{\text{Hydrolysis}}\mathrm{Fe(OH)}_3\downarrow+3\mathrm{HCl}\] (iron hydroxide sol)
2. Electrical Disintegration — Bredig's Arc Method
- An electric arc is struck between electrodes of the metal immersed in the dispersion medium
- The intense heat produces metal vapour that condenses to form colloidal particles
3. Peptization
-
A precipitate is converted into colloidal sol by shaking it with the dispersion medium in the presence of a small amount of electrolyte (the peptizing agent)
| Property Type | Property | Description |
|---|---|---|
| General Property | Nature | Heterogeneous system; particles pass slowly through membranes |
| Kinetic Property | Brownian Motion | Continuous random zig-zag motion of colloidal particles |
| Optical Property | Tyndall Effect | Scattering of light makes path of beam visible |
|
Colour |
Depends on wavelength of scattered light | |
| Electrical Property | Charge | Colloidal particles carry electric charge |
| Electrophoresis | Movement of particles towards oppositely charged electrode | |
| Electro-osmosis | Movement of dispersion medium under electric field | |
| Mechanical Property | Brownian Motion | Particles remain in constant motion |
| Stability Property | Coagulation | Precipitation of colloidal particles |
| Flocculation Value | ∝ 1 / Coagulating power |
1. Chemical Methods
-
Oxidation: \[\mathrm{SO}_2+2\mathrm{H}_2\mathrm{S}\xrightarrow{\mathrm{Oxidation}}3\mathrm{S}\downarrow+2\mathrm{H}_2\mathrm{O}\] (sulphur sol)
-
Reduction: \[2\mathrm{AuCl}_{3}+3\mathrm{HCHO}+3\mathrm{H}_{2}\mathrm{O}\xrightarrow{\mathrm{Reduction}}2\mathrm{Au}\downarrow+3\mathrm{HCOOH}+6\mathrm{HCl}\] (gold sol)
- Hydrolysis: \[\mathrm{FeCl}_3+3\mathrm{H}_2\mathrm{O}\xrightarrow{\text{Hydrolysis}}\mathrm{Fe(OH)}_3\downarrow+3\mathrm{HCl}\] (iron hydroxide sol)
2. Electrical Disintegration — Bredig's Arc Method
- An electric arc is struck between electrodes of the metal immersed in the dispersion medium
- The intense heat produces metal vapour that condenses to form colloidal particles
3. Peptization
-
A precipitate is converted into colloidal sol by shaking it with the dispersion medium in the presence of a small amount of electrolyte (the peptizing agent)
Emulsions show all properties of sols. Their particle size is 1000 Å to 10,000 Å; they scatter light (Tyndall effect).
Types of Emulsions:
| Feature | Oil in Water (O/W) | Water in Oil (W/O) |
|---|---|---|
| Dispersed phase | Oil | Water |
| Dispersion medium | Water | Oil |
| Continuous phase | Water | Oil |
| Miscibility | Addition of water mixes with it | Addition of oil mixes with it |
| Electrolyte effect | Small amount makes it conducting | No effect on conducting power |
| Emulsifiers used | Water-soluble alkali metal soaps, metal sulphates | Water-insoluble soaps (Zn, Al, Fe), alkaline earth metal soaps |
| Examples | Milk, vanishing cream | Butter, cream, cod liver oil |
Important Questions [65]
- Differentiate between the following: Adsorption and Absorption
- Give Reasons for the Following Observations : Nh3 Gas Adsorbs More Readily than N2 Gas on the Surface of Charcoal.
- Write three differences between physisorption and chemisorption.
- How Does Chemisorption Vary with Temperature?
- How Does Chemisorption Vary with Temperature?
- Give Reasons for the Following Observations : Powdered Substances Are More Effective Adsorbents.
- Give Reasons for the Following Observations: Physisorption Decreases with Increase in Temperature.
- Why is Adsorption Always Exothermic?
- Write the Differences Between Physisorption and Chemisorption with Respect to the Following
- Write One Similarity Between Physisorption and Chemisorption
- What is the Effect of Temperature on Chemisorption ?
- Physisorption is Reversible While Chemisorption is Irreversible. Why ?
- Differentiate Between Adsorbtion and Absorption.
- In Reference to Freundlich Adsorption Isotherm, Write the Expression for Adsorption of Gases on Solids in the Form of an Equation.
- Write One Difference in Homogeneous Catalysis and Heterogeneous Catalysis
- Write the Dispersed Phase and Dispersion Medium of the Following Colloidal Systems: (i) Smoke (ii) Milk
- It is necessary to remove CO when ammonia is prepared by Haber's process.
- Write the Dispersed Phase and Dispersion Medium of Butter.
- What is the Role of Promoters and Poisons in Catalysis?
- Co (G) and H2 (G) React to Give Different Products in the Presence of Different Catalysts. Which Ability of the Catalyst is Shown by These Reactions?
- Write One Difference in the Following: Multimolecular Colloid and Associated Colloid
- Define the Following with a Suitable Example, of Each: Gel
- Define the Following with a Suitable Example, of Each: Multimolecular Colloid
- What Type of Colloid is Formed When a Gas is Dispersed in a Liquid? Give an Example
- Write an Important Characteristic of Lyophilic Sols.
- Differentiate between the following: Multimolecular Colloid and Macromolecular colloid
- Give One Example Each of Lyophobic Sol and Lyophilic Sol.
- Write One Difference Between Multimolecular Colloid and Macromolecular Colloid
- Write One Difference Between Sol and Gel
- Write One Difference Between O/W Emulsion and W/O Emulsion
- Write One Difference In Lyophobic Sol and Lyophilic Sol
- Give One Example Each of Sol and Gel.
- Out of Sulphur Sol and Proteins, Which One Form Multimolecular Colloids?
- Write a Method by Which Lyophobic Colloids Can Be Coagulated.
- Define the following terms: Lyophilic colloid
- Give Reasons for the Following Observations: Lyophilic Sol is More Stable than Lyophobic Sol.
- Define the following term: Multimolecular colloids
- Differentiate between the following: Lyophobic Sol and Lyophilic Sol
- What Are Lyophilic and Lyophobic Colloids ? Which of These Sols Can Be Easily Coagulated on the Addition of Small Amounts of Electrolytes?
- Define the following term: Coagulation
- Give Reasons for the Following Observations: Leather Gets Hardened After Tanning.
- Out of BaCl2 and KCl, Which One is More Effective in Causing Coagulation of a Negatively Charged Colloidal Sol? Give Reason.
- Out of MgCl2 and AlCl3, which one is more effective in causing coagulation of negatively charged sol and why?
- What Happens When A Freshly Prepared Precipitate of Fe(Oh)3 is Shaken with a Small Amount of Fecl3 Solution?
- What Happens When Persistent Dialysis of a Colloidal Solution is Carried Out?
- What Happens When An Emulsion is Centrifuged?
- Give Reasons for the Following Observations: Brownian Movement Provides Stability to the Colloidal Solution.
- Define the Following Terms: Associated Colloids
- Define the following term: Zeta potential
- A Colloidal Sol is Prepared by the Given Method in Figure. What is the Charge on Hydrated Ferric Oxide Colloidal Particles Formed in the Test Tube? How is the Sol Represented?
- Define coagulation.
- State the Hardy-Schulze rule.
- What is electrophoresis?
- What is the Reason for the Stability of Colloidal Sols?
- Define the Following Term : Brownian Movement
- Why a negatively charged sol is obtained when AgNO3 solution is added to KI solution?
- Answer the Following Question. Why Are Medicines More Effective in the Colloidal State?
- A Colloidal Sol is Prepared by the Given Method in the Figure. What is the Charge on Hydrated Ferric Oxide Colloidal Particles Formed in the Test Tube? How is the Solution Represented?
- Answer the Following Question. What is the Difference Between an Emulsion and a Gel?
- Give One Example Each of 'Oil in Water' and 'Water in Oil' Emulsion.
- Give Reasons for the Following Observations: Addition of Alum Purifies the Water
- What are emulsions?
- What Are the Dispersed Phase and Dispersion Medium in Milk?
- What are the different types emulsions? Give one example of each type.
- Based on the Type of Particles of Dispersed Phase, Give One Example Each of Associated Colloid and Multimolecular Colloid.
Concepts [21]
- Introduction of Adsorption
- Distinction Between Adsorption and Absorption
- Mechanism of Adsorption
- Types of Adsorption
- Adsorption Isotherms (Freundlich and Langmuir Adsorption Isotherm)
- Adsorption from Solution Phase
- Applications of Adsorption
- Homogeneous and Heterogeneous Catalysis
- Adsorption Theory of Heterogeneous Catalysis
- Shape-selective Catalysis by Zeolites
- Enzyme Catalysis
- Catalysts in Industry
- Colloids
- Preparation of Colloids
- Purification of Colloidal Solution
- Properties of Colloidal Dispersions
- Preparation of Colloids
- Purification of Colloidal Solution
- Properties of Colloidal Solutions
- Emulsions
- Colloids Around Us
