Advertisements
Advertisements
Question
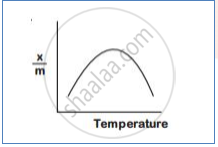
How does chemisorption vary with temperature?
Advertisements
Solution
With an increase in temperature chemisorption first increases as molecules get activation energy for adsorption. After certain chemisorption decreases.temperature,
APPEARS IN
RELATED QUESTIONS
Physisorption is reversible while chemisorption is irreversible. Why ?
Give reasons for the following observations :
Powdered substances are more effective adsorbents.
Answer in one sentence:
Name type of adsorption in which the compound is formed.
Which of the following is an example of physisorption?
In physisorption, adsorption is generally ____________.
The physical adsorption is due to ____________.
In physisorption adsorbent does not show specificity for any particular gas because ______.
Why is it important to have clean surface in surface studies?
In physical adsorption, gas molecules are bound on the solid surface by
Write three differences between physisorption and chemisorption.
