Advertisements
Advertisements
प्रश्न
How does chemisorption vary with temperature?
Advertisements
उत्तर
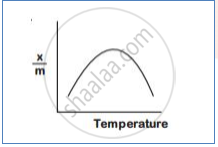
With an increase in temperature chemisorption first increases as molecules get activation energy for adsorption. After certain chemisorption decreases.temperature,
APPEARS IN
संबंधित प्रश्न
Differentiate between adsorbtion and absorption.
What is the effect of temperature on chemisorption ?
What is the difference between physisorption and chemisorption?
Discuss the effect of temperature on the adsorption of gases on solids.
Physisorption is reversible while chemisorption is irreversible. Why ?
Which of the following is true about chemisorption?
Chromatography is based on the principle of ____________.
The extent of adsorption increases with the ____________.
Which of the following is not a favourable condition for physical adsorption?
In physical adsorption, gas molecules are bound on the solid surface by
