English Medium
Academic Year: 2025-2026
Date & Time: 25th February 2026, 10:30 am
Duration: 3h
Advertisements
General Instructions:
Read the following instructions carefully and follow them:
- This question paper contain 39 questions. Allquestions are compulsory.
- The question paper is divided into three sections - A, B and C.
Section A: Biology (30 marks)
Section B: Chemistry (25 marks)
Section C: Physics (25 marks) - The question paper has MCQs, VSAs, SAs, LAs and CBQs. Marks are given against each question.
- There are case based questions (CBQs) with three sub-questions and are of 4 marks each.
- Divide your answer sheet into three sections as per the question paper -
Section A (Biology), Section B (Chemistry) and Section C (Physics). It is compulsory to answer each question in its respective section. Do not mix answers of one section into the other section. - Instructions are given with each section and question, wherever necessary.
- Kindly note that a separate question paper has been provided for visually impaired candidates.
- There is no overall choice in the question paper. However, an internal choice has been provided in a few questions. Only one of the choices in such questions
must be attempted.
Observe the figure given below and identify the incorrect step for the activity set-up:
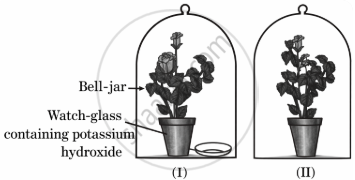
Both plants are kept in the dark room for three days before placing them in separate glass jars.
Both plants are covered with separate wooden bell-jars.
Use vaseline to seal the bottom of the jars with the glass plates.
Both potted plants were kept in sunlight for a few hours before the iodine test for the presence of starch.
Chapter:
Organisms of a higher trophic level which feed on several types of organisms belonging to a number of lower trophic level form the ______.
Food web
Ecosystem
Ecological pyramid
Food chain
Chapter:
In a synapse, the chemical signal is transmitted from ______.
dendritic end of one neuron to axonal end of another neuron.
axon to cell body of the same neuron.
cell body to axonal end of the same neuron.
axonal end of one neuron to dendritic end of another neuron.
Chapter: [6] Control and Co-ordination
The growth of tendrils in grapes plants is due to ______.
Effect of light.
Effect of gravity.
Rapid cell division in tendril cells in contact with the support.
Rapid cell division in tendril cells that are away from the support.
Chapter:
In accumulation of toxic substances at higher trophic levels of an ecosystem through the food chain, which of the following organisms is affected the most?
Small aquatic plants
Human beings
Small fish
Large fish
Chapter:
In plants, the role of cytokinin is to/in:
promote cell division
wilting of leaves
promote opening of stomatal pore
growth of stem
Chapter:
The number of chromosomes in parents and offspring of a particular species undergoing sexual reproduction remains constant due to ______.
doubling of chromosomes after zygote formation.
halving of chromosomes after zygote formation.
doubling of chromosomes before gamete formation.
halving of chromosomes at the time of gamete formation.
Chapter:
Assertion (A): The basic filtration unit in the kidneys, like in the lungs, is a cluster of very thin-walled blood capillaries.
Reason (R): Each capillary cluster in the kidney is associated with the cup-shaped end of a coiled tube called Bowman’s capsule.
Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of Assertion (A).
Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of Assertion (A).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false, but Reason (R) is true.
Chapter:
Assertion (A): Ozone (O3) gas at higher levels of atmosphere is a product of UV radiation acting on Oxygen (O2).
Reason (R): The amount of ozone in the atmosphere began to droр sharply in 1980s.
Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of Assertion (A).
Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of Assertion (A).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false, but Reason (R) is true.
Chapter:
What kind of respiration provides more energy and why?
Chapter:
Separation of right side and left side of the heart is useful to keep oxygenated have different and deoxygenated blood from mixing. Various organisms types of heart.
(I) Fill in the blanks (i) to (iii) correctly according to the type of organisms and their evolution in terms of blood circulation:
| Organisms | Number of chambers in heart | Type of blood being circulated | Type of circulation |
| Fish | (i) ______ | Mixed | Single |
| (ii) ______ | 3-chambered | Mixed | Incomplete double circulation |
| Birds and Mammals | 4-chambered | Separated circulation path of oxygenated and deoxygenated blood | (iii) ______ |
(II) Which type of circulation is most efficient?
Chapter:
In the following food chain, 5 J of energy is available to man. Calculate, how much energy was available at the producer level. Why?
Plant → Goat → Man
Chapter:
A blue colour flower (BB) bearing plant is crossed with a plant with white coloured flowers (bb).
- State the colour of the flowers you would expect in their F1 progeny.
- What must be the percentage of white flower plants in the F2 generation, if the F1 plants are self-pollinated?
- State the expected ratio of genotypes BB and bb in F2 progeny.
Chapter:
In xylem tissue, vessels and tracheids of the roots, stems and leaves are interconnected to form a continuous system of water-conducting channels reaching all parts of the plant. Explain how this is achieved.
Chapter:
Read the following passage and answer the questions that follow:
| Not all plants carry out photosynthesis by the same mechanism. In most plants, it depends directly on gaseous carbon dioxide that diffuses into the leaf. However, some plants can store carbon dioxide in the vacuole of the leaf cells as part of a complex carbon compound. This compound is transported to the chloroplast and releases carbon dioxide when required for photosynthesis. This special mechanism is believed to have evolved as an adaptation in such plants which do not open stomata during the daytime to conserve water for survival in dry conditions (desert conditions). |
Attempt either (a) or (b):
-
- Which process in these plants is minimised to help the plants survive in dry conditions?
- Which part of the leaf is responsible for exchange of gases during photosynthesis?
OR
- List the events that occur during the process of photosynthesis.
- When are such desert plants likely to take in carbon dioxide from the environment, unlike other plants?
- Draw a labelled diagram of stomata with open stomatal pore.
Chapter:
Observe the given diagram and answer the questions that follow:
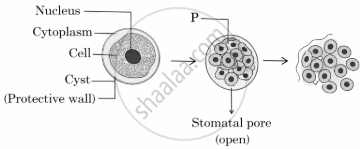
- Give the name and type of reproduction shown in the above diagram.
- Comment on the processes taking place in stage P.
- Name an organism which reproduces by the above mentioned method.
- Name the method that can be used to raise plants that have lost their capacity to produce seeds. List any two advantages of growing plants by such a method.
Chapter:
Observe the given diagram and answer the questions that follow:
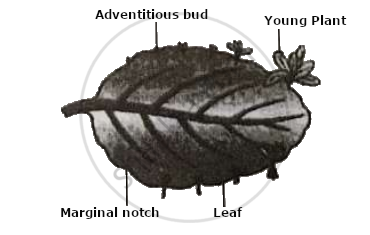
- Give the name of the structure and type of reproduction shown in the above diagram.
- State one advantage and one disadvantage of the above mentioned method of reproduction.
- Give reason: Asexual reproduction lacks genetic variation.
- Why have higher organisms resorted to sexual reproduction in spite of its complexity?
Chapter:
When zinc reacts with NaOH, the product formed is ______.
Na2O
ZnO
Na2ZnO2
Zn(OH)2
Chapter:
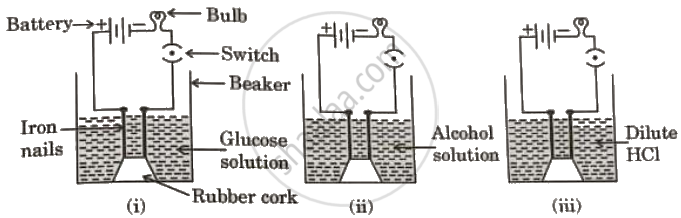
In which case/cases will the bulb glow considering that all other connections of electric circuit are complete and working?
Case (i) only
Case (ii) only
Cases (ii) and (iii)
Case (iii) only
Chapter:
The reaction of calcium with cold water is ______.
Less violent, but the heat evolved is sufficient for the hydrogen to catch fire.
Very violent and exothermic and hydrogen catches fire.
Exothermic, but the heat evolved is not sufficient for the hydrogen to catch fire.
Slow and endothermic.
Chapter:
Advertisements
Which of the following compounds will undergo addition reaction?
C4H10
C4H8
C3H8
CH4
Chapter:
In the following equation
\[\ce{x KI + H2O2 -> I2 + y KOH}\]
x and y respectively are:
2, 2
1, 2
2, 1
1, 1
Chapter:
To a small amount of copper oxide in a beaker, when we add dilute hydrochloric acid slowly with stirring, the change in the colour of the solution is due to the formation of ______.
Copper (II) oxide, which is black in colour.
Copper (I) chloride, which is blue in colour.
Copper (II) chloride, which is blue-green in colour.
Copper (I) oxide, which is black in colour.
Chapter:
Which of the following is an example of a displacement reaction?
The combustion of natural gas.
The reaction of a reactive metal with dilute acid.
The electrolysis of water.
The reaction of quick lime with water.
Chapter:
Assertion (A): There are large number of compounds with many carbon atoms linked to each other.
Reason (R): Carbon-Carbon bond is very strong and stable.
Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of Assertion (A).
Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of Assertion (A).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false, but Reason (R) is true.
Chapter:
Comment on the following property of ionic compounds and give a reason in support of your answer:
Physical nature
Chapter:
Comment on the following property of ionic compounds and give reason in support of your answer:
Melting and boiling points
Chapter:
Give reason:
Potato chips manufacturers fill the packet of chips with nitrogen gas.
Chapter:
Give reason:
Respiration is an exothermic reaction.
Chapter:
Why do we store silver chloride in a dark coloured bottle?
Chapter: [1] Chemical Reactions and Equations
While diluting an acid, why is it recommended that the acid should be added to water and not water to the acid?
Chapter:
Give reason:
Dry HCl gas does not turn blue litmus red whereas dilute hydrochloric acid does.
Chapter:
Give reason:
Blue colour of Copper(II) sulphate crystals turns white on heating.
Chapter:
Write the preparation of the following compound with balanced chemical equation:
NaOH by chlor-alkali process
Chapter:
Write the preparation of the following compound with balanced chemical equation:
Baking Soda
Chapter:
Write the preparation of the following compound with balanced chemical equation:
Plaster of Paris
Chapter:
Read the following passage and answer the questions that follow:
|
An electrochemical process is used for refining metals by selectively dissolving and depositing them through electrolysis. Electrolytic refining is used for metals like Copper, Zinc, Tin, Nickel, Silver and Gold. |
- Define anode mud. [1]
- In electrolytic refining of copper, name the anode and the cathode. [1]
- With the help of chemical equations, show the extraction of copper from its ore Cu2S. [2]
OR - Draw a neat and labelled diagram showing electrolytic refining of copper. [2]
- With the help of chemical equations, show the extraction of copper from its ore Cu2S. [2]
Chapter:
What is homologous series?
Chapter: [4] Carbon and its Compounds
Which two of the following organic compounds belong to the same homologous series?
C2H4, C4H10, C3H4, C4H6
Chapter:
Write the product(s) in the following reaction:
\[\ce{CH3 - CH2 - OH + CH3COOH ->[Acid]}\]
Chapter:
Advertisements
Write the product(s) in the following reaction:
\[\ce{C2H5OH + O2 ->[Burning]}\]
Chapter:
Why does micelle formation take place when soap is added to water?
Chapter:
Name the following compound:
\[\begin{array}{cc}
\ce{CH3 - C - CH3}\\
||\\
\ce{O}\\
\end{array}\]
Chapter:
Name the following compound:
\[\begin{array}{cc}
\ce{H}\phantom{..............}\\
|\phantom{..............}\\
\ce{H - C - C ≡ C - H}\\
|\phantom{..............}\\
\ce{H}\phantom{..............}\\
\end{array}\]
Chapter:
Name the reaction which is commonly used in the conversion of vegetable oils to fats.
Chapter:
Why is the melting point of C4H10 higher than C3H8?
Chapter:
What happens when a small piece of sodium is dropped in ethanol? Write the equation for this reactions.
Chapter: [4] Carbon and its Compounds
Write the products in the following reaction:
\[\ce{CH3COOC2H5 + NaOH ->}\]
Chapter:
Observe the given ray diagram showing refraction through a prism and choose the correct option for colour of rays 1 and 2:
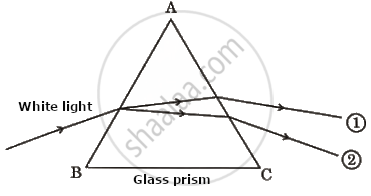
Colour of ray 1 : Blue, Colour of ray 2 : Red
Colour of ray 1 : Green, Colour of ray 2 : Yellow
Colour of ray 1 : Red, Colour of ray 2 : Violet
Colour of ray 1 : Violet, Colour of ray 2 : Red
Chapter:
In an electric circuit carrying current I, a 2 Ω resistor is replaced by an 8 Ω resistor without changing the connected (ideal) battery. Then ______.
current becomes `I/4` and potential difference `V/4`.
current becomes `I/4` and potential difference remains V.
current becomes 4I and potential difference remains V.
current becomes 4I and potential difference `V/4`.
Chapter:
Assertion (A): The needle of a compass, kept in an external magnetic field, always aligns itself in the North-South direction of the Earth.
Reason (R): The behaviour of the needle of the compass is similar to the behaviour of a freely suspended bar magnet.
Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of Assertion (A).
Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of Assertion (A).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false, but Reason (R) is true.
Chapter:
What happens when light is scattered from very fine particles of medium? Justify your answer in given case.
Chapter:
What happens when light is scattered from very large size particles of medium? Justify your answer in given case.
Chapter:
With the help of a ray diagram, explain any phenomenon of formation of spectrum in nature.
Chapter:
Redraw the following ray diagram in your answer book and show the path of reflected/refracted ray:
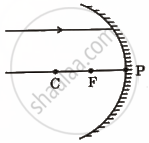
Chapter:
Redraw the following ray diagram in your answer book and show the path of reflected/refracted ray:
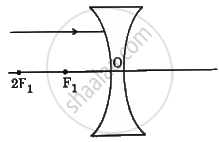
Chapter:
Name a device which is used to maintain a constant potential difference in a circuit.
Chapter:
Name a device which is used to change the electric current in an electric circuit.
Chapter:
When the potential difference between the terminals of an electric heater is 110 V, a current of 5 A flows through it. What will be the value of current flowing through it when the potential difference is increased to 220 V?
Chapter:
Consider the given electric circuit:
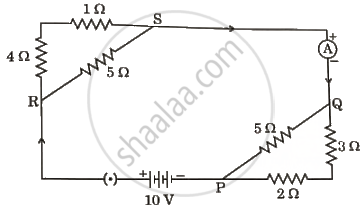
Calculate the following:
- Total resistance of the circuit.
- The electric current drawn from the battery.
- Potential difference between points P and Q.
Chapter:
Study the given diagram and answer the questions that follow:

- Write the name of the eye defect shown in the diagram. Where is the image formed in this eye defect with respect to the retina of the eye?
- List two causes of this eye defect.
- With the help of a ray diagram, show how this defect of vision is corrected.
Chapter:
Other Solutions
Submit Question Paper
Help us maintain new question papers on Shaalaa.com, so we can continue to help studentsonly jpg, png and pdf files
CBSE previous year question papers Class 10 Science with solutions 2025 - 2026
Previous year Question paper for CBSE Class 10 Science-2026 is solved by experts. Solved question papers gives you the chance to check yourself after your mock test.
By referring the question paper Solutions for Science, you can scale your preparation level and work on your weak areas. It will also help the candidates in developing the time-management skills. Practice makes perfect, and there is no better way to practice than to attempt previous year question paper solutions of CBSE Class 10.
How CBSE Class 10 Question Paper solutions Help Students ?
• Question paper solutions for Science will helps students to prepare for exam.
• Question paper with answer will boost students confidence in exam time and also give you an idea About the important questions and topics to be prepared for the board exam.
• For finding solution of question papers no need to refer so multiple sources like textbook or guides.
