Advertisements
Advertisements
Question
Read the following passage and answer the questions that follow:
|
An electrochemical process is used for refining metals by selectively dissolving and depositing them through electrolysis. Electrolytic refining is used for metals like Copper, Zinc, Tin, Nickel, Silver and Gold. |
- Define anode mud. [1]
- In electrolytic refining of copper, name the anode and the cathode. [1]
-
- With the help of chemical equations, show the extraction of copper from its ore Cu2S. [2]
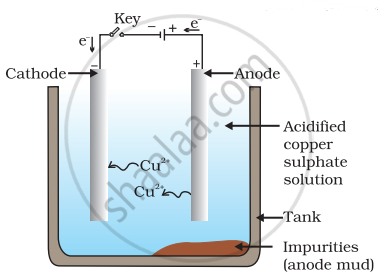
OR - Draw a neat and labelled diagram showing electrolytic refining of copper. [2]
- With the help of chemical equations, show the extraction of copper from its ore Cu2S. [2]
Chemical Equations/Structures
Definition
Diagram
Short Answer
Advertisements
Solution
a. Anode mud is the insoluble impurities that settle down at the bottom of the electrolytic cell during electrolytic refining. These impurities fall from the anode and collect below it.
b. Anode: Impure copper rod
Cathode: Thin sheet of pure copper
c. i. Copper is extracted from copper glance (Cu2S) by the following steps:
Roasting: \[\ce{2Cu2S + 3O2 -> 2Cu2O + 2SO2}\]
Self-reduction: \[\ce{2Cu2O + Cu2S -> 6Cu + SO2}\]
OR
ii.
shaalaa.com
Is there an error in this question or solution?
2025-2026 (March) 31/3/2
