Advertisements
Advertisements
Questions
Write the reaction involved in the preparation of phenol from benzene sulphonic acid.
How will you convert the following? (Write a chemical equation.)
Phenol from benzene sulphonic acid.
Advertisements
Solution

APPEARS IN
RELATED QUESTIONS
Answer in brief.
Give the equations of the reactions for the preparation of phenol from isopropyl benezene.
Answer in brief.
Give a simple chemical test to distinguish between ethanol and ethyl bromide.
An ether (A), C5H12O, when heated with excess of hot HI produce two alkyl halides which on hydrolysis form compound (B) and (C), oxidation of (B) gave and acid (D), whereas oxidation of (C) gave a ketone (E). Deduce the structural formula of (A), (B), (C), (D), and (E).
Reaction between hot conc. HI and anisole gives ______________
Write Kolbe’s reaction.
How will you bring about the following conversions?
isopropyl alcohol to acetone
Write chemical reactions to convert phenol into salicyaldehyde. Write the name of the reaction. What happens when if CCl4 is used instead of CHCl3 in the above reaction.
Phenol reacts with concentrated nitric acid in the presence of cone. H2SO4 to form ____________.
Number of oxygen atoms present in salicylaldehyde are ______.
Which of the following reagents are required in the conversion of phenol to salicylic acid?
Which of the following compounds does not react with bromine in alkaline medium?
Sodium metal with ethyl alcohol gives __________ gas.
The CORRECT decreasing order of boiling points for isomeric primary (1°), secondary (2°) and tertiary (3°) alcohols is ____________.
Which of following elements does not react with hot concentrated sulphuric acid?
Identify 'A' and 'B' in the following series of reactions.
The product 'C' in the following reaction is:
\[\ce{CH3CH2Br ->[alc. KCN] {'A'} ->[H3O^+][\Delta] {'B'} ->[i. LiAlH4][ii. H3O^+] {'C'}}\]
Which of the following is INCORRECT regarding phenol?
Which isomer of C4H10O is optically active?
Which following reagent is used to obtain alkene from alcohol?
Which of the following alcohols is least soluble in water?
Identify 'Z' in the following series of reaction:
\[\ce{Butan - 2 - ol ->[PCl3] X ->[alco. KOH] Y ->[i) H2SO4][ii) H-OH/heat] Z}\]
Identify the alcohol that reacts immediately with Lucas reagent?
The compound which reacts fastest with Lucas reagent at room temperature is ______.
Name the catalyst used in commercial method of preparation of phenol.

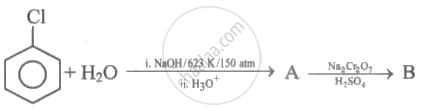
Product (B) in this reaction is:
What is the product obtained in Reimer - Tiemann reaction?
The major product obtained in the following reaction is

Write the product when 1°, 2° and 3° alcohol vapours are passed over hot copper.
Write chemical reaction when hot copper is treated with Vapours of 3° (tertiary) alcohol.
Convert the following :
cumene to phenol.
