Advertisements
Advertisements
Question
What is the action of following reagents on pent-3-enal?
- H2/Ni
- LiAlH4/H3O+
Advertisements
Solution
Pent-3-enal contains an isolated > C = C < and an aldehyde group. H2/Ni can reduce both these functional groups while LiAlH4 can reduce only –CHO of the two.
- H2/Ni:
\[\ce{\underset{\text{Pent-3-enal}}{CH3 - CH = CH - CH2 - CHO} ->[H2/Ni]\underset{\text{Pentan-1-ol}}{CH3 - CH2 - CH2 - CH2 - CH2 - OH}}\] - LiAlH4/H3O+:
\[\ce{\underset{\text{Pent-3-enal}}{CH3 - CH = CH - CH2 - CHO} ->[(i)LiAIH4][(ii)H3O+] \underset{\text{Pent-3-en-1-ol}}{CH3 - CH = CH - CH2 - CH2 - OH}}\]
RELATED QUESTIONS
Answer in brief.
Explain why phenol is more acidic than ethyl alcohol.
Answer in brief.
Give the reagents and conditions necessary to prepare phenol from Benzene sulfonic acid.
An ether (A), C5H12O, when heated with excess of hot HI produce two alkyl halides which on hydrolysis form compound (B) and (C), oxidation of (B) gave and acid (D), whereas oxidation of (C) gave a ketone (E). Deduce the structural formula of (A), (B), (C), (D), and (E).
Name the reagents used to convert phenol into
- picric acid
- p-benzoquinone
An unknown alcohol is treated with Lucas reagent. Explain how you will determine whether the alcohol is primary, secondary or tertiary. Indicate by chemical equation the reaction between isopropyl alcohol and Lucas reagent.
What is the action of conc. H2SO4 on carbolic acid at 373 K.
Which alcohol is difficult to oxidise?
Sodium metal with ethyl alcohol gives __________ gas.
Isobutylene on hydroboration followed by oxidation with hydrogen peroxide in presence of base yields ______.
Propane when treated with cold cone. H2SO4 forms a compound which on heating with water gives ______.
The number of isomeric alcohols possible with the formula C4H10O is ____________.
\[\ce{CH3 - CH = CH2 ->[HBr][Peroxide] A}\]
Identify A.
Carbolic acid is oxidised by acidified sodium dichromate to give ______.
In Dow process, chlorobenzene is converted into phenol by ____________.
+I effect of alkyl groups in alcohols increases the stability of ____________.
Sodium benzene sulphonate reacts with NaOH and then on acidic hydrolysis, it gives __________.
\[\ce{Isopropyl alcohol + acidic K2Cr2O7 -> X}\]
Identify product 'X' in the above reactions.
The number of moles of hydrogen gas formed when 2 moles of 2-methylpropan-2-ol reacts with aluminium is ____________.
Which of the following alcohols is least soluble in water?
Identify the compound having highest boiling point from the following?
Identify the product X in the following reaction.
\[\ce{Phenol ->[Na2Cr2O7][H2SO4] X}\]
What is the product formed when aniline is treated with \[\ce{NaNO2 + HCl}\] previous to hydrolysis?
What is the product obtained in Reimer - Tiemann reaction?
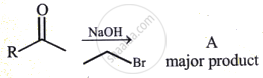
The major product obtained in the following reaction is

Arrange O - H, C - H and N - H bonds in increasing order of their bond polarity.
Explain: Phenols are acid while alcohol is neutral.
The structure of A in the given reaction is: