Advertisements
Advertisements
प्रश्न
What is the action of following reagents on pent-3-enal?
- H2/Ni
- LiAlH4/H3O+
Advertisements
उत्तर
Pent-3-enal contains an isolated > C = C < and an aldehyde group. H2/Ni can reduce both these functional groups while LiAlH4 can reduce only –CHO of the two.
- H2/Ni:
\[\ce{\underset{\text{Pent-3-enal}}{CH3 - CH = CH - CH2 - CHO} ->[H2/Ni]\underset{\text{Pentan-1-ol}}{CH3 - CH2 - CH2 - CH2 - CH2 - OH}}\] - LiAlH4/H3O+:
\[\ce{\underset{\text{Pent-3-enal}}{CH3 - CH = CH - CH2 - CHO} ->[(i)LiAIH4][(ii)H3O+] \underset{\text{Pent-3-en-1-ol}}{CH3 - CH = CH - CH2 - CH2 - OH}}\]
संबंधित प्रश्न
Answer in one sentence/ word.
Write the name of the electrophile used in Kolbe’s Reaction.
Answer in brief.
Explain why phenol is more acidic than ethyl alcohol.
Answer in brief.
Give the reagents and conditions necessary to prepare phenol from Benzene sulfonic acid.
When vapours of tert.butyl alcohol are passed over hot copper, it gives _____________
Write IUPAC name of crotonyl alcohol.
Write chemical equation of acetyl chloride with phenol
What is the action of following on phenol at low temperature?
- dil. HNO3
- conc. H2SO4
- Br2/CS2
What is the action of conc. H2SO4 on carbolic acid at 373 K.
Write chemical reactions to convert phenol into salicyaldehyde. Write the name of the reaction. What happens when if CCl4 is used instead of CHCl3 in the above reaction.
Phenol reacts with concentrated nitric acid in the presence of cone. H2SO4 to form ____________.
In the Lucas test for alcohols, the appearance of turbidity is due to the formation of ____________.
Which of the following reagents are required in the conversion of phenol to salicylic acid?
Which of following elements does not react with hot concentrated sulphuric acid?
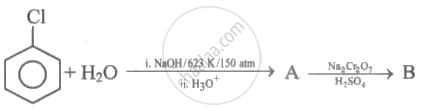
Identify 'A' and 'B' in the following series of reactions.
\[\ce{CH3 - CH = CH2 ->[HBr][Peroxide] A}\]
Identify A.
Which of the following alcohols has tertiary allylic carbon?
In Raschig's method for synthesis of phenol, the reactants used are ____________.
The number of moles of hydrogen gas formed when 2 moles of 2-methylpropan-2-ol reacts with aluminium is ____________.
Which isomer of C4H10O is optically active?
A reaction of phenol with chloroform in presence of sodium hydroxide to form salicylaldehyde is known as ____________.
Which of the following alcohols is NOT prepared by reduction of carbonyl compounds?
Identify the product X in the following reaction.
\[\ce{Phenol ->[Na2Cr2O7][H2SO4] X}\]
Which among the following phenolic compound is most acidic in nature?

Product (B) in this reaction is:
What is the product formed when aniline is treated with \[\ce{NaNO2 + HCl}\] previous to hydrolysis?
Which among the following is not the method of preparation of phenol?
The major product obtained in the following reaction is

Write the chemical reaction when hot copper is treated with Vapours of 1° (primary) alcohol.
Write the chemical reaction when hot copper is treated with Vapours of 2° (secondary) alcohol.
Propene can be converted into propan-1-ol by:
