Advertisements
Advertisements
Question
Convert the following :
cumene to phenol.
Advertisements
Solution

APPEARS IN
RELATED QUESTIONS
Answer in brief.
Give the reagents and conditions necessary to prepare phenol from Benzene sulfonic acid.
Reaction between hot conc. HI and anisole gives ______________
Write chemical equation of acetyl chloride with phenol
An unknown alcohol is treated with Lucas reagent. Explain how you will determine whether the alcohol is primary, secondary or tertiary. Indicate by chemical equation the reaction between isopropyl alcohol and Lucas reagent.
How will you bring about the following conversions?
acetone to 2-methylpropan-2-ol
Write the reactions for the preparation of carbolic acid from aniline.
An organic compound gives hydrogen on reaction with sodium metal. It forms an aldehyde with molecular formula C2H4O on oxidation with pyridinium chlorochromate. Give the chemical equations in support of these observations.
Which of the following compounds does not react with bromine in alkaline medium?
α-butylene when subjected to hydroboration oxidation reaction, yields ______.
Phenol is obtained from cumene ____________.
Which of following elements does not react with hot concentrated sulphuric acid?
Identify 'A' and 'B' in the following series of reactions.
Phenoxide ion is more stable than phenol due to the ____________.
In Raschig's method for synthesis of phenol, the reactants used are ____________.
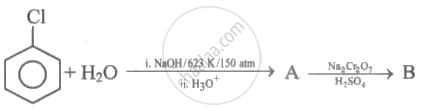
Identify the product B in following conversion.
\[\ce{Chlorobenzene + H2O ->[Cu, 673 K][Pressure] A ->[conc. H2SO4][373 K] B}\]
Which isomer of C4H10O is optically active?
Which following reagent is used to obtain alkene from alcohol?
Which among the following compounds is used to prepare solid fuel using calcium acetate?
Which among the following is allylic secondary alcohol?
Which of the following on oxidation yields ethyl methyl ketone?
Which of the following alcohols is NOT prepared by reduction of carbonyl compounds?
Identify the product obtained when phenol is treated with bromine water?
Identify the product X in the following reaction.
\[\ce{Phenol ->[Na2Cr2O7][H2SO4] X}\]
Which among the following phenolic compound is most acidic in nature?
Name the catalyst used in commercial method of preparation of phenol.
Which of the following compounds reacts immediately with Lucas reagent?
What is the product obtained in Reimer - Tiemann reaction?
What is the action of nitrous acid on aniline?
Write chemical reaction when hot copper is treated with Vapours of 3° (tertiary) alcohol.
Which of the following is trihydric phenol?
