Advertisements
Advertisements
Question
Advertisements
Solution
Within a group the element with the largest size is expected to be present at the bottom of the group.
APPEARS IN
RELATED QUESTIONS
Answer the following in respect of element `31/15 P`
Give its electronic configuration
Answer the following in respect of element `31/15 P `
Give its formula with chlorine?
Give the trends in atomic size on moving across the period left to right.
Give reasons for the following:
The size of the anion is greater than the size of the parent atom.
Give scientific reasons for the following:
Atomic radius goes on decreasing while going from left to right in a period.
Write a scientific reason.
Atomic radius goes on increasing down a group.
Some elements and their atomic radii are given here. Arrange them in decreasing order of their atomic radii. Identify which of the above elements is the biggest atom and which is smallest?
| Element | K | Na | Rb | Cs | Li |
| Atomic radius (pm) | 231 | 186 | 244 | 262 | 151 |
Carbon belongs to the second period and Group 14. Silicon belongs to the third period and Group 14. If the atomic number of carbon is 6, the atomic number of silicon is ______
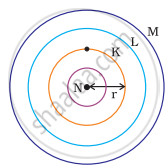
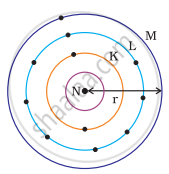
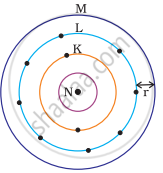
Which one of the following depict the correct representation of atomic radius(r) of an atom?
|
(i) |
(ii) |
|
(iii) |
(iv) |
Arrange the following in order of increasing radii:
Mg2+, Mg, Mg+
Explain your choice.