Advertisements
Advertisements
Question
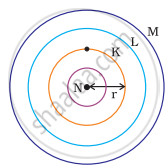
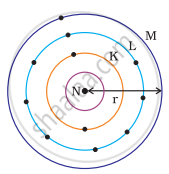
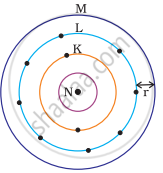
Which one of the following depict the correct representation of atomic radius(r) of an atom?
|
(i) |
(ii) |
|
(iii) |
(iv) |
Options
(i) and (ii)
(ii) and (iii)
(iii) and (iv)
(i) and (iv)
MCQ
Advertisements
Solution
(ii) and (iii)
Explanation -
The atomic radius(r) of an atom is the distance between the centre of the nucleus and the outermost shell of an isolated atom. In representation (ii), the outermost shell is K and atomic radius is correctly depicted from centre of nucleus to shell K while in representation (iii), the outermost shell is M and atomic radius is correctly depicted from centre of nucleus to shell M.
shaalaa.com
Is there an error in this question or solution?