Advertisements
Advertisements
Question
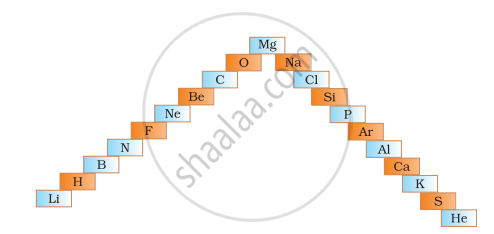
Give the trends in atomic size on moving across the period left to right.
Advertisements
Solution
Atomic size decreases from left to right as the Atomic Number increases by one at each stage, with one proton added to the nucleus and one electron added to the valence shell. As nuclear attraction rises, the size of the atom decreases.
RELATED QUESTIONS
Answer the following in respect of element `31/15 P `
Give its formula with chlorine?
Give the trends in atomic size on moving down the group.
Arrange the following in order of increasing radii:
CI- , CI
Fill in the blanks.
Size of the atoms ______ from left to right across a period and ______ on descending in a group of normal elements.
Arrange the following in increasing order of property indicated
I, I+, I- (atomic size)
The following questions refer to the Periodic Table.
What happens to the atomic size of elements moving from top to bottom of a group?
Nanometer unit is used to measure atomic radius.
An element X has mass number 40 and contains 21 neutrons in its atom. To which group of the Periodic Table does it belong?
Write the formula of the product formed when the element A (atomic number 19) combines with the element B (atomic number 17). Draw its electronic dot structure. What is the nature of the bond formed?
- In below ladder symbols of elements are jumbled up. Rearrange these symbols of elements in the increasing order of their atomic number in the Periodic Table.
- Arrange them in the order of their group also.