Advertisements
Advertisements
Question
Why does NH3 form hydrogen bond but PH3 does not?
Advertisements
Solution 1
Nitrogen is highly electronegative as compared to phosphorus. This causes a greater attraction of electrons towards nitrogen in NH3 than towards phosphorus in PH3. Hence, the extent of hydrogen bonding in PH3 is very less as compared to NH3.
Solution 2
Nitrogen has an electronegativity value of 3.0, which is much higher than that of H (2.1). As a result, N-H bond is quite polar and hence NH3 undergoes intermolecular H-bonding.
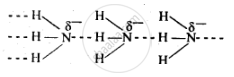
Phosphorus has an electronegativity value of 2.1. Thus, the P–H bond is not polar, and hence PH3 does not undergo H-bonding.
RELATED QUESTIONS
Account for the following: Oxygen shows catenation behavior less than sulphur.
Give reasons: SO2 is reducing while TeO2 is an oxidising agent.
Give reasons for the following : H2Te is the strongest reducing agent amongst all the hydrides of Group 16 elements.
Why is H2O a liquid and H2S a gas?
Which of the following does not react with oxygen directly?
Zn, Ti, Pt, Fe
The HNH angle value is higher than HPH, HAsH and HSbH angles. Why? [Hint: Can be explained on the basis of sp3 hybridisation in NH3 and only s−p bonding between hydrogen and other elements of the group].
Justify the placement of O, S, Se, Te and Po in the same group of the periodic table in terms of electronic configuration, oxidation state and hydride formation.
Knowing the electron gain enthalpy values for \[\ce{O -> O-}\] and \[\ce{O -> O^{2-}}\] as −141 and 702 kJ mol−1 respectively, how can you account for the formation of a large number of oxides having O2− species and not O−?
(Hint: Consider lattice energy factor in the formation of compounds).
Arrange the following in the order of the property indicated against set :
H2O, H2S, H2Se, H2Te − increasing acidic character.
Give a reason for the following:
Fluorine gives only one oxide but chlorine gives a series of oxides.
Arrange the following in order of the property indicated set.
HF, HCl, HBr, HI - decreasing bond enthalpy.
Which of the following statement is incorrect?
Match the items of Columns I and II and mark the correct option.
| Column I | Column II |
| (A) \[\ce{H2SO4}\] | (1) Highest electron gain enthalpy |
| (B) \[\ce{CCl3NO2}\] | (2) Chalcogen |
| (C) \[\ce{Cl2}\] | (3) Tear gas |
| (D) Sulphur | (4) Storage batteries |
Strong reducing behaviour of \[\ce{H3PO2}\] is due to ______.
Which of the following statements are correct?
(i) \[\ce{CaF2 + H2SO4 -> CaSO4 + 2HF}\]
(ii) \[\ce{2HI + H2SO4 -> I2 + SO2 + 2H2O}\]
(iii) \[\ce{Cu + 2H2SO4 -> CuSO4 + SO2 + 2H2O}\]
(iv) \[\ce{Nacl + H2SO4 -> NaHSO4 + HCl}\]
Out of \[\ce{H2O}\] and \[\ce{H2S}\], which one has higher bond angle and why?
In forming (i) \[\ce{N2 -> N^{+}2}\] and (ii) \[\ce{O2 -> O^{+}2}\]; the electrons respectively are removed from:
What is the basicity of \[\ce{H3PO4}\]?
______ is a radioactive element in group 16 elements.
