Advertisements
Advertisements
प्रश्न
Why does NH3 form hydrogen bond but PH3 does not?
Advertisements
उत्तर १
Nitrogen is highly electronegative as compared to phosphorus. This causes a greater attraction of electrons towards nitrogen in NH3 than towards phosphorus in PH3. Hence, the extent of hydrogen bonding in PH3 is very less as compared to NH3.
उत्तर २
Nitrogen has an electronegativity value of 3.0, which is much higher than that of H (2.1). As a result, N-H bond is quite polar and hence NH3 undergoes intermolecular H-bonding.
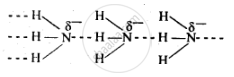
Phosphorus has an electronegativity value of 2.1. Thus, the P–H bond is not polar, and hence PH3 does not undergo H-bonding.
संबंधित प्रश्न
Give reasons: SO2 is reducing while TeO2 is an oxidising agent.
Account for the following : There is large difference between the melting and boiling points of oxygen and sulphur.
Give reasons for the following : H2Te is the strongest reducing agent amongst all the hydrides of Group 16 elements.
The HNH angle value is higher than HPH, HAsH and HSbH angles. Why? [Hint: Can be explained on the basis of sp3 hybridisation in NH3 and only s−p bonding between hydrogen and other elements of the group].
Why are halogens strong oxidising agents?
Arrange the following in the order of property indicated for the given set:
F2, Cl2, Br2, I2 - increasing bond dissociation enthalpy.
Give reasons Thermal stability decreases from H2O to H2Te.
Draw the structures of `H_3PO_2`
Explain the following properties of group 16 elements :
1) Electro negativity
2) Melting and boiling points
3) Metallic character
4) Allotropy
Give reactions for the following:
O – O single bond is weaker than S – S single bond.
Give a reason for the following:
Fluorine gives only one oxide but chlorine gives a series of oxides.
Given below are two statements labelled as Assertion (A) and Reason (R).
Assertion (A): Electron gain enthalpy of oxygen is less than that of Flourine but greater than Nitrogen.
Reason (R): Ionisation enthalpies of the elements follow the order Nitrogen > Oxygen > Fluorine.
Select the most appropriate answer from the options given below:
Strong reducing behaviour of \[\ce{H3PO2}\] is due to ______.
Which of the following statements are correct?
(i) \[\ce{CaF2 + H2SO4 -> CaSO4 + 2HF}\]
(ii) \[\ce{2HI + H2SO4 -> I2 + SO2 + 2H2O}\]
(iii) \[\ce{Cu + 2H2SO4 -> CuSO4 + SO2 + 2H2O}\]
(iv) \[\ce{Nacl + H2SO4 -> NaHSO4 + HCl}\]
Write a balanced chemical equation for the reaction showing catalytic oxidation of NH3 by atmospheric oxygen.
Out of \[\ce{H2O}\] and \[\ce{H2S}\], which one has higher bond angle and why?
In forming (i) \[\ce{N2 -> N^{+}2}\] and (ii) \[\ce{O2 -> O^{+}2}\]; the electrons respectively are removed from:
______ is a gaseous element of group 16.
Given below are two statements:
Statement I: The boiling point of hydrides of Group 16 elements follows the order:
H2O > H2Te > H2Se > H2S
Statement II: On the basis of molecular mass, H2O is expected to have a lower boiling point than the other members of the group but due to the presence of extensive H-bonding in H2O, it has a higher boiling point.
In the light of the above statements, choose the correct answer from the options given below:
