Advertisements
Advertisements
Questions
Why are aryl halides less reactive towards nucleophilic substitution reactions than alkyl halides?
Why are aryl halides less reactive than alkyl halides towards nucleophilic substitution reactions?
Why are haloarenes less reactive towards nucleophilic substitution reactions as compared to haloalkanes?
Advertisements
Solution
Aryl halides are less reactive towards nucleophilic substitution reaction due to the following reasons.
- In haloarenes, the lone pair of electron on halogen are in resonance with benzene ring. So, C – Cl bond acquires partial double bond character which strengthen C – Cl bond and difficult to be substituted by nucleophile.
Therefore, they are less reactive towards nucleophilic substitution reaction.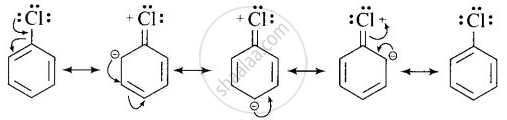
- In haloarenes, the carbon atom attached to halogen is sp2 hybridised. The sp2 hybridised carbon is more electronegative than sp3 hybridised carbon. This sp2-hybridised carbon in haloarenes can hold the electron pair of \[\ce{C - X}\] bond more tightly and make this \[\ce{C - Cl}\] bond shorter than \[\ce{C Cl}\] bond of haloalkanes.
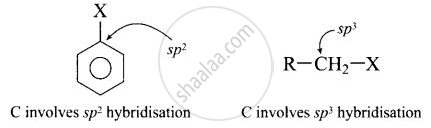
Since, it is difficult to break a shorter bond than a longer bond, therefore, halorenes are less reactive than haloalkanes. - In haloarenesm the phenyl cation is not stabilised by resonance therefore SN1 mechanism cannot be followed.
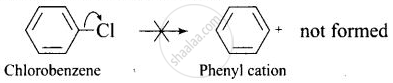
- Because of the repulsion between the nucleophile and electron-rich arenas, aryl halides are less reactive than alkyl halides.
APPEARS IN
RELATED QUESTIONS
How do you convert the following:
Ethanol to propanenitrile
Out of  , which is more reactive towards SN1 reaction and why?
, which is more reactive towards SN1 reaction and why?
Which would undergo SN2 reaction faster in the following pair and why ?

Given reasons: SN1 reactions are accompanied by racemization in optically active alkyl halides.
Which of the following is optically inactive?
Halogenation of alkanes is ____________.
Racemic compound has ____________.
The increasing order of nucleophilicity would be:
Which of the following compound will undergo racemisation when reacts with aq. KOH?
(i)
(ii)
CH3CH2CH2Cl
(iii)
\[\begin{array}{cc}
\ce{CH3}\phantom{..}\\
|\phantom{....}\\
\ce{CH3-CH-CH2Cl}
\end{array}\]
(iv)
\[\begin{array}{cc}
\phantom{..}\ce{H}\\
\phantom{..}|\\
\ce{CH3-C-Cl}\\
\phantom{..}|\\
\phantom{.....}\ce{C2H5}
\end{array}\]
Ethylene chloride and ethylidene chloride are isomers. Identify the correct statements.
(i) Both the compounds form same product on treatment with alcoholic KOH.
(ii) Both the compounds form same product on treatment with aq.NaOH.
(iii) Both the compounds form same product on reduction.
(iv) Both the compounds are optically active.
Which of the compounds will react faster in SN1 reaction with the –OH ion?
\[\ce{CH3-CH2-Cl}\] or \[\ce{C6H5-CH2-Cl}\]
Write the structures and names of the compounds formed when compound ‘A’ with molecular formula, \[\ce{C7H8}\] is treated with \[\ce{Cl2}\] in the presence of \[\ce{FeCl3}\].
Match the reactions given in Column I with the types of reactions given in Column II.
| Column I | Column II | |
| (i) | 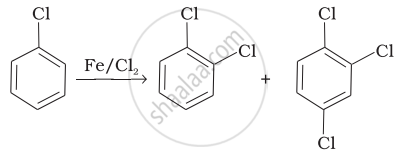 |
(a) Nucleophilic aromatic substitution |
| (ii) | \[\begin{array}{cc} \ce{CH3 - CH = CH2 + HBr -> CH3 - CH - CH3}\\ \phantom{............................}|\phantom{}\\ \phantom{.............................}\ce{Br}\phantom{} \end{array}\] |
(b) Electrophilic aromatic substitution |
| (iii) | 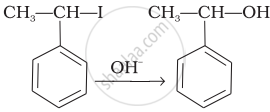 |
(c) Saytzeff elimination |
| (iv) | 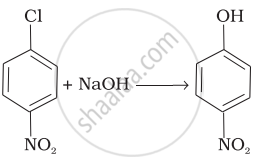 |
(d) Electrophilic addition |
| (v) | \[\begin{array}{cc} \ce{CH3 CH2 CH CH3 ->[alc.KOH] CH3 CH = CH CH3}\\ \phantom{}|\phantom{..........................}\\ \phantom{}\ce{Br}\phantom{........................} \end{array}\] |
(e) Nucleophilic substitution (SN1) |
Which one of the following compounds is more reactive towards SN1 reaction?
Give reason for the following:
The product formed during SN1 reaction is a racemic mixture.
The number of chiral alcohol (s) with molecular formula C4H10O is ______.
In SN1 reactions, the correct order of reactivity for the following compounds:
CH3Cl, CH3CH2Cl, (CH3)2CHCl and (CH3)3CCl is ______.
Retention of configuration is observed in ______.
Give the mechanism of the following reaction:
\[\ce{CH3CH2OH ->[H2SO4][413 K] CH3CH2-O-CH2CH3 + H2O}\]
Which alkyl halide from the following pair would you expect to react more rapidly by an SN2 mechanism? Explain your answer.
\[\begin{array}{cc}\ce{CH3CHCH2CH2Br}\\|\phantom{.........}\\\ce{CH3}\phantom{......}\end{array}\] or \[\begin{array}{cc}\ce{CH3CH2CHCH2Br}\\\phantom{}|\\\phantom{...}\ce{CH3}\end{array}\]
