Advertisements
Advertisements
Question
Which of the following statements are in accordance with the Arrhenius equation?
(i) Rate of a reaction increases with increase in temperature.
(ii) Rate of a reaction increases with decrease in activation energy.
(iii) Rate constant decreases exponentially with increase in temperature.
(iv) Rate of reaction decreases with decrease in activation energy.
Advertisements
Solution
(i) Rate of a reaction increases with increase in temperature.
(ii) Rate of a reaction increases with decrease in activation energy.
Explanation:
The rate of reaction increases with temperature. It becomes double with every 10° change. It increases with decrease in activation energy.
APPEARS IN
RELATED QUESTIONS
Consider the reaction
`3I_((aq))^-) +S_2O_8^(2-)->I_(3(aq))^-) + 2S_2O_4^(2-)`
At particular time t, `(d[SO_4^(2-)])/dt=2.2xx10^(-2)"M/s"`
What are the values of the following at the same time?
a. `-(d[I^-])/dt`
b. `-(d[S_2O_8^(2-)])/dt`
c. `-(d[I_3^-])/dt`
The rate constant of a first order reaction increases from 2 × 10−2 to 4 × 10−2 when the temperature changes from 300 K to 310 K. Calculate the energy of activation (Ea).
(log 2 = 0.301, log 3 = 0.4771, log 4 = 0.6021)
The activation energy for the reaction \[\ce{2 HI_{(g)} -> H2_{(g)} + I2_{(g)}}\] is 209.5 kJ mol−1 at 581 K. Calculate the fraction of molecules of reactants having energy equal to or greater than activation energy?
The rate of a reaction quadruples when the temperature changes from 293 K to 313 K. Calculate the energy of activation of the reaction assuming that it does not change with temperature.
A first-order reaction is 50% completed in 40 minutes at 300 K and in 20 minutes at 320 K. Calculate the activation energy of the reaction. (Given : log 2 = 0·3010, log 4 = 0·6021, R = 8·314 JK–1 mol–1)
Which of the following graphs represents exothermic reaction?
(a)

(b)
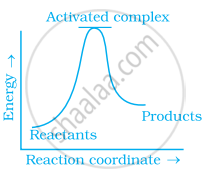
(c)
Why does the rate of a reaction increase with rise in temperature?
Oxygen is available in plenty in air yet fuels do not burn by themselves at room temperature. Explain.
Why in the redox titration of \[\ce{KMnO4}\] vs oxalic acid, we heat oxalic acid solution before starting the titration?
Total number of vibrational degrees of freedom present in CO2 molecule is
For an endothermic reaction energy of activation is Ea and enthalpy of reaction ΔH (both of there in KJ moI–1) minimum value of Ea will be ______.
The activation energy in a chemical reaction is defined as ______.
The activation energy in a chemical reaction is defined as ______.
Explain how and why will the rate of reaction for a given reaction be affected when the temperature at which the reaction was taking place is decreased.
A first-order reaction is 50% complete in 30 minutes at 300 K and in 10 minutes at 320 K. Calculate activation energy (Ea) for the reaction. [R = 8.314 J K−1 mol−1]
[Given: log 2 = 0.3010, log 3 = 0.4771, log 4 = 0.6021]
It is generally observed that the rate of a chemical reaction becomes double with every 10°C rise in temperature. If the generalisation holds true for a reaction in the temperature range of 298 K to 308 K, what would be the value of activation energy (Ea) for the reaction?
The rate of a reaction quadruples when temperature changes from 27°C to 57°C calculate the energy of activation.
(Given: R = 8. 314 J K−1 mol−1, log 4 = 0.6021)
Given below are two statements:
Statement I: The nutrient deficient water bodies lead to eutrophication.
Statement II: Eutrophication leads to decrease in the level of oxygen in the water bodies.
In the light of the above statements, choose the correct answer from the options given below:
