Advertisements
Advertisements
Question
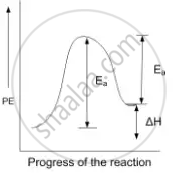
For an endothermic reaction energy of activation is Ea and enthalpy of reaction ΔH (both of there in KJ moI–1) minimum value of Ea will be ______.
Options
Less than ΔH
Equal to ΔH
More than ΔH
Equal to zero
MCQ
Fill in the Blanks
Advertisements
Solution
For an endothermic reaction energy of activation is Ea and enthalpy of reaction ΔH (both of there in KJ moI–1) minimum value of Ea will be less than ΔH.
Explanation:
For an endothermic reaction
ΔH = Eaf – Eab
Eaf = Activation energy for forward reaction
Eab = Activation energy for backward reaction
or Eaf = ΔH + Eab
So Ea > ΔH
shaalaa.com
Is there an error in this question or solution?
