Advertisements
Advertisements
Question
Just as precise measurements are necessary in science, it is equally important to be able to make rough estimates of quantities using rudimentary ideas and common observations. Think of ways by which you can estimate the following (where an estimate is difficult to obtain, try to get an upper bound on the quantity):-
the number of air molecules in your classroom.
Advertisements
Solution 1
Let the volume of the room be V.
One mole of air at NTP occupies 22.4 l i.e., 22.4 × 10–3 m3 volume.
Number of molecules in one mole = 6.023 × 1023
∴Number of molecules in room of volume V
=`(6.023xx10^(23))/(22.4xx10^(-3))xxV`
= 134.915 × 1026 V
= 1.35 × 1028 V
Solution 2
We can determine the volume of the class-room by measuring its length, breadth and height. Consider a class room of size 10 m x 8 m x 4 m.
Volume of this room is 320 m3.
We know that 22.4l or 22.4 x 10-3 m3 of air has 6.02 x 1023 molecules (equal to Avogadro’s number).
Number of molecules of air in the class room =(6.02 x 1023 /22.4 x 10-3 ) x 320 =8.6 x 1027
RELATED QUESTIONS
Exercise
Solve the numerical example.
When the planet Jupiter is at a distance of 824.7 million kilometers from the Earth, its angular diameter is measured to be 35.72'' of arc. Calculate the diameter of Jupiter.
Length of one’s chest can be measured using metre scale.
Complete the analogy.
Height of a person: cm :: Length of your sharpened pencil lead:______?
Match the following.
| 1. | Length of the fore arm | metre |
| 2. | SI unit of length | second |
| 3. | Nano | 103 |
| 4. | SI Unit of time | 10–9 |
| 5. | Kilo | Cubit |
Rulers, measuring tapes and metre scales are used to measure ______
The fencing of a square garden is 20 m in length. How long is one side of the garden?
Chumki, Jhumri, and Imran need wire to make a fence.

- How much wire in all will the three need?
There are two beautiful lakes near a village. People come for boating and picnics in both the lakes. The village Panchayat is worried that with the noise of the boats the birds will stop coming. The Panchayat wants motorboats in only one lake. The other lake will be saved for the birds to make their nests.
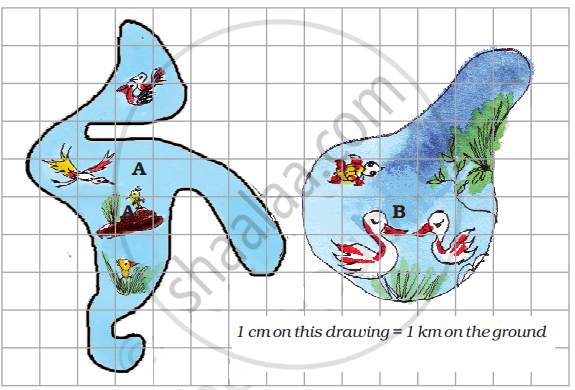
- What is the length of the boundary of lake B in the drawing?
There are two beautiful lakes near a village. People come for boating and picnics in both the lakes. The village Panchayat is worried that with the noise of the boats the birds will stop coming. The Panchayat wants motorboats in only one lake. The other lake will be saved for the birds to make their nests.

- A longer boundary around the lake will help more birds to lay their eggs. So which lake should be kept for birds? Which lake should be used for boats?
In which SI unit, you can measure your height?
What formula is used to measure the area of your classroom?
What is the unit of measurements of very small lengths?
The radius of atom is of the order of 1 Å and the radius of nucleus is of the order of fermi. How many magnitudes higher is the volume of atom as compared to the volume of nucleus?
Why is the speed of light used to define the metre?
One parsec is defined as ______.
The parallax method is used to measure astronomical distances because ______.
Which instruments are used to measure very small distances at the atomic scale?
