Advertisements
Advertisements
Question
The rate constant for the decomposition of N2O5 at various temperatures is given below:
| T/°C | 0 | 20 | 40 | 60 | 80 |
| 105 × k/s−1 | 0.0787 | 1.70 | 25.7 | 178 | 2140 |
Draw a graph between ln k and `1/T` and calculate the values of A and Ea. Predict the rate constant at 30º and 50ºC.
Advertisements
Solution
The rate constants for the decomposition of N2O5 at different temperatures are shown below.
| T(°C) | T(K) | 1/T | k(s−1) | In k (= 2.303 log k) |
| 0 | 273 | 3.6 × 10−3 | 7.87 × 10−7 | −14.06 |
| 20 | 293 | 3.4 × 10−3 | 1.70 × 10−5 | −10.98 |
| 40 | 313 | 3.19 × 10−3 | 25.7 × 10−5 | −8.266 |
| 60 | 333 | 3.00 × 10−3 | 178 × 10−5 | −6.332 |
| 80 | 353 | 2.8 × 10−3 | 2140 × 10−5 | −3.844 |
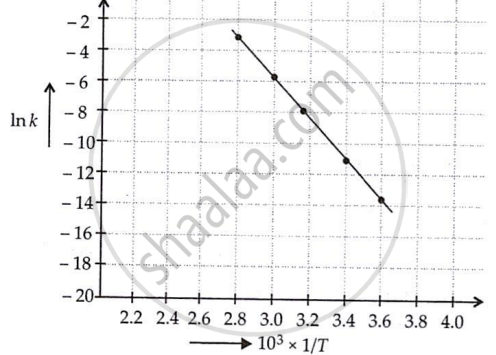
Slope of the line = tan θ
= `(y_2 - y_1)/(x_2 - x_1)`
= `(-10.98 - (-14.06))/(3.4 - 3.6) xx 10^3`
= `3.08/-0.2 xx 10^3`
= −15.4 × 103
Ea = −slope × R
= −(−15.4 × 103 × 8.314)
= 128.035 kJ K−1 mol−1
Again In A = In k + `E_a/(RT)`
= `-14.06 + (128.035 xx 10^3)/(8.314 xx 273)`
= `-14.06 + 128035/2269.722`
= −14.06 + 56.40
= 42.34
or, log A = 42.34
or, A = antilog 42.34 = 0.3388 × 1019
or, A = 3.3888 × 1018
Values of rate constant k at 303 K and 323 K can be obtained from the graph.
First, k is obtained corresponding to `1/(303 K) and 1/(323 K)` and then k is calculated.
APPEARS IN
RELATED QUESTIONS
Explain a graphical method to determine activation energy of a reaction.
(b) Rate constant ‘k’ of a reaction varies with temperature ‘T’ according to the equation:
`logk=logA-E_a/2.303R(1/T)`
Where Ea is the activation energy. When a graph is plotted for `logk Vs. 1/T` a straight line with a slope of −4250 K is obtained. Calculate ‘Ea’ for the reaction.(R = 8.314 JK−1 mol−1)
The rate constant for the first-order decomposition of H2O2 is given by the following equation:
`logk=14.2-(1.0xx10^4)/TK`
Calculate Ea for this reaction and rate constant k if its half-life period be 200 minutes.
(Given: R = 8.314 JK–1 mol–1)
Consider a certain reaction \[\ce{A -> Products}\] with k = 2.0 × 10−2 s−1. Calculate the concentration of A remaining after 100 s if the initial concentration of A is 1.0 mol L−1.
The decomposition of A into product has value of k as 4.5 × 103 s−1 at 10°C and energy of activation 60 kJ mol−1. At what temperature would k be 1.5 × 104 s−1?
The rate constant of a first order reaction are 0.58 S-1 at 313 K and 0.045 S-1 at 293 K. What is the energy of activation for the reaction?
Calculate activation energy for a reaction of which rate constant becomes four times when temperature changes from 30 °C to 50 °C. (Given R = 8.314 JK−1 mol−1).
Write a condition under which a bimolecular reaction is kinetically first order. Give an example of such a reaction. (Given : log2 = 0.3010,log 3 = 0.4771, log5 = 0.6990).
Which of the following graphs represents exothermic reaction?
(a)

(b)
(c)
During decomposition of an activated complex:
(i) energy is always released
(ii) energy is always absorbed
(iii) energy does not change
(iv) reactants may be formed
Why does the rate of a reaction increase with rise in temperature?
Total number of vibrational degrees of freedom present in CO2 molecule is
For an endothermic reaction energy of activation is Ea and enthalpy of reaction ΔH (both of there in KJ moI–1) minimum value of Ea will be ______.
In respect of the eqn k = \[\ce{Ae^{{-E_a}/{RT}}}\] in chemical kinetics, which one of the following statement is correct?
The activation energy in a chemical reaction is defined as ______.
Arrhenius equation can be represented graphically as follows:

The (i) intercept and (ii) slope of the graph are:
The equation k = `(6.5 xx 10^12 "s"^(-1))"e"^(- 26000 " K"//"T")` is followed for the decomposition of compound A. The activation energy for the reaction is ______ kJ mol-1. (Nearest integer) (Given: R = 8.314 JK-1 mol-1)
The decomposition of N2O into N2 and O2 in the presence of gaseous argon follows second-order kinetics, with k = (5.0 × 1011 L mol−1 s−1) `"e"^(-(29000 "K")/"T")`. Arrhenius parameters are ______ kJ mol−1.
Which plot of ln k vs `1/T` is consistent with the Arrhenius equation?
