Advertisements
Advertisements
Question
Consider a certain reaction \[\ce{A -> Products}\] with k = 2.0 × 10−2 s−1. Calculate the concentration of A remaining after 100 s if the initial concentration of A is 1.0 mol L−1.
Advertisements
Solution 1
Given: k = 2.0 × 10−2 s−1,
T1 = 0,
[A]1 = 1.0 M,
T2 = 100 s,
[A]2 = ?
k = `1/(T_2 - T_1) ln ([A]_1)/([A]_2)`
2.0 × 10−2 = `1/(100 - 0) ln 1.0/([A]_2)`
(2.0 × 10−2) × 100 = `ln 1.0/([A]_2)`
2 = `ln 1/([A]_2)`
7.389 = `1/([A]_2)`
∴ [A]2 = `1/7.389`
= 0.135 mol L−1
Solution 2
The units of k (s−1) indicate that the reaction is of first order. For a first order reaction,
k = `2.303/t log_10 [A]_0/([A])`
∴ 2.0 × 10−2 = `2.303/100 log_10 1.0/([A])`
= `-2.303/100 log_10 [A]`
or, log10 [A] = `-(2.0 xx 10^-2 xx 100)/2.303`
or, log10 [A] = `-2/2.303`
or, log10 [A] = −0.8684
or, [A] = antilog10 (−0.8684)
[A] = 0.1354 mol L−1
APPEARS IN
RELATED QUESTIONS
Explain a graphical method to determine activation energy of a reaction.
Consider the reaction
`3I_((aq))^-) +S_2O_8^(2-)->I_(3(aq))^-) + 2S_2O_4^(2-)`
At particular time t, `(d[SO_4^(2-)])/dt=2.2xx10^(-2)"M/s"`
What are the values of the following at the same time?
a. `-(d[I^-])/dt`
b. `-(d[S_2O_8^(2-)])/dt`
c. `-(d[I_3^-])/dt`
The rate constant for the decomposition of N2O5 at various temperatures is given below:
| T/°C | 0 | 20 | 40 | 60 | 80 |
| 105 × k/s−1 | 0.0787 | 1.70 | 25.7 | 178 | 2140 |
Draw a graph between ln k and `1/T` and calculate the values of A and Ea. Predict the rate constant at 30º and 50ºC.
The rate constant for the decomposition of hydrocarbons is 2.418 × 10−5 s−1 at 546 K. If the energy of activation is 179.9 kJ/mol, what will be the value of pre-exponential factor?
The decomposition of hydrocarbon follows the equation k = `(4.5 xx 10^11 s^-1) e^(-28000 K//T)`
Calculate Ea.
The rate of a reaction quadruples when the temperature changes from 293 K to 313 K. Calculate the energy of activation of the reaction assuming that it does not change with temperature.
In the Arrhenius equation for a first order reaction, the values of ‘A’ of ‘Ea’ are 4 × 1013 sec−1 and 98.6 kJ mol−1 respectively. At what temperature will its half life period be 10 minutes?
[R = 8.314 J K−1 mol−1]
What is the effect of adding a catalyst on Activation energy (Ea)
Write a condition under which a bimolecular reaction is kinetically first order. Give an example of such a reaction. (Given : log2 = 0.3010,log 3 = 0.4771, log5 = 0.6990).
Predict the main product of the following reactions: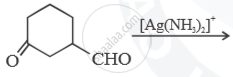
Oxygen is available in plenty in air yet fuels do not burn by themselves at room temperature. Explain.
Match the statements given in Column I and Column II
| Column I | Column I | |
| (i) | Catalyst alters the rate of reaction | (a) cannot be fraction or zero |
| (ii) | Molecularity | (b) proper orientation is not there always |
| (iii) | Second half life of first order reaction | (c) by lowering the activation energy |
| (iv) | `e^((-E_a)/(RT)` | (d) is same as the first |
| (v) | Energetically favourable reactions (e) total probability is one are sometimes slow | (e) total probability is one |
| (vi) | Area under the Maxwell Boltzman curve is constant | (f) refers to the fraction of molecules with energy equal to or greater than activation energy |
In respect of the eqn k = \[\ce{Ae^{{-E_a}/{RT}}}\] in chemical kinetics, which one of the following statement is correct?
The activation energy in a chemical reaction is defined as ______.
Arrhenius equation can be represented graphically as follows:

The (i) intercept and (ii) slope of the graph are:
Explain how and why will the rate of reaction for a given reaction be affected when the temperature at which the reaction was taking place is decreased.
The activation energy of one of the reactions in a biochemical process is 532611 J mol–1. When the temperature falls from 310 K to 300 K, the change in rate constant observed is k300 = x × 10–3 k310. The value of x is ______.
[Given: ln 10 = 2.3, R = 8.3 J K–1 mol–1]
A schematic plot of ln Keq versus inverse of temperature for a reaction is shown below

The reaction must be:
What happens to the rate constant k and activation energy Ea as the temperature of a chemical reaction is increased? Justify.
