Advertisements
Advertisements
प्रश्न
The rate constant for the decomposition of N2O5 at various temperatures is given below:
| T/°C | 0 | 20 | 40 | 60 | 80 |
| 105 × k/s−1 | 0.0787 | 1.70 | 25.7 | 178 | 2140 |
Draw a graph between ln k and `1/T` and calculate the values of A and Ea. Predict the rate constant at 30º and 50ºC.
Advertisements
उत्तर
The rate constants for the decomposition of N2O5 at different temperatures are shown below.
| T(°C) | T(K) | 1/T | k(s−1) | In k (= 2.303 log k) |
| 0 | 273 | 3.6 × 10−3 | 7.87 × 10−7 | −14.06 |
| 20 | 293 | 3.4 × 10−3 | 1.70 × 10−5 | −10.98 |
| 40 | 313 | 3.19 × 10−3 | 25.7 × 10−5 | −8.266 |
| 60 | 333 | 3.00 × 10−3 | 178 × 10−5 | −6.332 |
| 80 | 353 | 2.8 × 10−3 | 2140 × 10−5 | −3.844 |
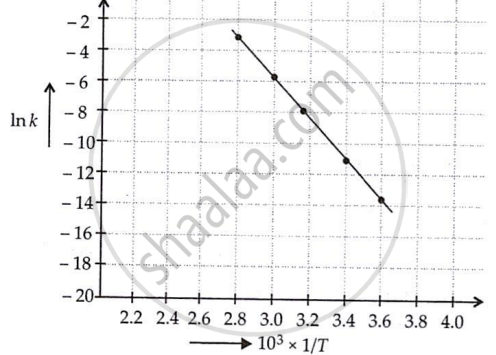
Slope of the line = tan θ
= `(y_2 - y_1)/(x_2 - x_1)`
= `(-10.98 - (-14.06))/(3.4 - 3.6) xx 10^3`
= `3.08/-0.2 xx 10^3`
= −15.4 × 103
Ea = −slope × R
= −(−15.4 × 103 × 8.314)
= 128.035 kJ K−1 mol−1
Again In A = In k + `E_a/(RT)`
= `-14.06 + (128.035 xx 10^3)/(8.314 xx 273)`
= `-14.06 + 128035/2269.722`
= −14.06 + 56.40
= 42.34
or, log A = 42.34
or, A = antilog 42.34 = 0.3388 × 1019
or, A = 3.3888 × 1018
Values of rate constant k at 303 K and 323 K can be obtained from the graph.
First, k is obtained corresponding to `1/(303 K) and 1/(323 K)` and then k is calculated.
APPEARS IN
संबंधित प्रश्न
The rate constant for the first-order decomposition of H2O2 is given by the following equation:
`logk=14.2-(1.0xx10^4)/TK`
Calculate Ea for this reaction and rate constant k if its half-life period be 200 minutes.
(Given: R = 8.314 JK–1 mol–1)
The rate constant of a first order reaction increases from 2 × 10−2 to 4 × 10−2 when the temperature changes from 300 K to 310 K. Calculate the energy of activation (Ea).
(log 2 = 0.301, log 3 = 0.4771, log 4 = 0.6021)
The activation energy for the reaction \[\ce{2 HI_{(g)} -> H2_{(g)} + I2_{(g)}}\] is 209.5 kJ mol−1 at 581K. Calculate the fraction of molecules of reactants having energy equal to or greater than activation energy?
The rate of a reaction quadruples when the temperature changes from 293 K to 313 K. Calculate the energy of activation of the reaction assuming that it does not change with temperature.
Calculate activation energy for a reaction of which rate constant becomes four times when temperature changes from 30 °C to 50 °C. (Given R = 8.314 JK−1 mol−1).
What is the effect of adding a catalyst on Activation energy (Ea)
A first-order reaction is 50% completed in 40 minutes at 300 K and in 20 minutes at 320 K. Calculate the activation energy of the reaction. (Given : log 2 = 0·3010, log 4 = 0·6021, R = 8·314 JK–1 mol–1)
The decomposition of a hydrocarbon has value of rate constant as 2.5×104s-1 At 27° what temperature would rate constant be 7.5×104 × 3 s-1if energy of activation is 19.147 × 103 J mol-1 ?
The reaction between \[\ce{H2(g)}\] and \[\ce{O2(g)}\] is highly feasible yet allowing the gases to stand at room temperature in the same vessel does not lead to the formation of water. Explain.
Why in the redox titration of \[\ce{KMnO4}\] vs oxalic acid, we heat oxalic acid solution before starting the titration?
The rate constant for a reaction is 1.5 × 10–7 sec–1 at 50°C. What is the value of activation energy?
The activation energy in a chemical reaction is defined as ______.
Explain how and why will the rate of reaction for a given reaction be affected when the temperature at which the reaction was taking place is decreased.
The activation energy of one of the reactions in a biochemical process is 532611 J mol–1. When the temperature falls from 310 K to 300 K, the change in rate constant observed is k300 = x × 10–3 k310. The value of x is ______.
[Given: ln 10 = 2.3, R = 8.3 J K–1 mol–1]
The equation k = `(6.5 xx 10^12 "s"^(-1))"e"^(- 26000 " K"//"T")` is followed for the decomposition of compound A. The activation energy for the reaction is ______ kJ mol-1. (Nearest integer) (Given: R = 8.314 JK-1 mol-1)
The decomposition of N2O into N2 and O2 in the presence of gaseous argon follows second-order kinetics, with k = (5.0 × 1011 L mol−1 s−1) `"e"^(-(29000 "K")/"T")`. Arrhenius parameters are ______ kJ mol−1.
A first-order reaction is 50% complete in 30 minutes at 300 K and in 10 minutes at 320 K. Calculate activation energy (Ea) for the reaction. [R = 8.314 J K−1 mol−1]
[Given: log 2 = 0.3010, log 3 = 0.4771, log 4 = 0.6021]
What happens to the rate constant k and activation energy Ea as the temperature of a chemical reaction is increased? Justify.
Which plot of ln k vs `1/T` is consistent with the Arrhenius equation?
