Advertisements
Advertisements
Question
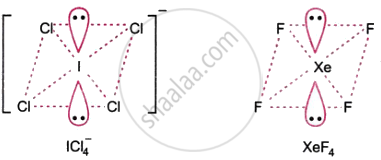
Give the formula and describe the structure of a noble gas species which is isostructural with:
\[\ce{ICI^-_4}\]
Advertisements
Solution
ICl4 is isostructural with XeF4 as both are square planar.
RELATED QUESTIONS
Account for the following:
Helium is used in diving apparatus.
Draw the structures of the following molecules: XeF2
What happens when: XeF4 reacts with SbF5?
Write the structures of the following molecule:
XeOF4
Complete the following equation:
\[\ce{XeF2 + H2O ->}\]
Balance the following equation:
\[\ce{XeF6 + H2O -> XeO2F2 + HF}\]
How are xenon fluorides XeF2, XeF4 and XeF6 obtained?
Give the formula and describe the structure of a noble gas species which is isostructural with:
\[\ce{IBr^-_2}\]
Give the formula and describe the structure of a noble gas species which is isostructural with:
\[\ce{BrO^-_3}\]
Write electronic configuration and two uses of neon. (Z = 10)
Write the electronic configuration of the following element:
Krypton (Z = 36)
Sulfur dioxide reacts with sodium hydroxide solution to form _______.
The number of lone pairs of electrons present in ClF5:
Which of the following reactions is an example of redox reaction?
On partial hydrolysis, XeF6 gives ______.
Match List - I with List - II:
| List - I | List - II | ||
| (Species) | (Number of lone pairs of electrons on the central atom) |
||
| (A) | XeF2 | (i) | 0 |
| (B) | XeO2F2 | (ii) | 1 |
| (C) | XeO3F2 | (iii) | 2 |
| (D) | XeF4 | (iv) | 3 |
Choose the most appropriate answer from the options given below:
Discuss the trends in electronegativity and atomic radii for elements of group 16 17, 18.
