Advertisements
Advertisements
Question
Label the one conjugate acid-base pair in the following reaction.
\[\ce{CO^{2-}_{3(aq)} + H2O_{(l)} ⇌ OH^- + HCO^-_3}\]
Advertisements
Solution
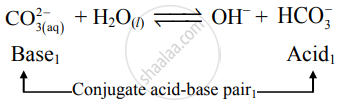
APPEARS IN
RELATED QUESTIONS
Answer the following in brief :
What are acids and bases according to Arrhenius theory?
Answer the following in brief :
What is meant by conjugate acid-base pair?
Label the conjugate acid-base pair in the following reaction:
\[\ce{HCl + H2O ⇌ H3O\overset{\oplus} + Cl\overset{\ominus}{}}\]
Label the conjugate acid-base pair in the following reaction:
\[\ce{CO^2-_3 + H2O ⇌ OH- + HCO^-_3}\]
Answer the following in brief :
Write a reaction in which water acts as a base.
The theory which explain amphoteric nature of water is ______.
The conjugate acid of \[\ce{HSO^-_4}\] is ____________.
Which among the following compounds is used as selective weed killer?
In which oxidation state, group 15 elements act as Lewis base?
Which of the following fluro compounds is most likely to behave as a Lewis base?
Which of the following can act as Lowry – Bronsted acid as well as base?
Discuss the Lowry – Bronsted concept of acids and bases.
Identify the conjugate acid-base pair for the following reaction in an aqueous solution.
\[\ce{HS^-_{( aq)} + HF ⇌ F^-_{( aq)} + H2S_{(aq)}}\]
Identify the conjugate acid-base pair for the following reaction in an aqueous solution.
\[\ce{HPO^{2-}_4 + SO^{2-}_3 ⇌ PO^{3-}_4 + HSO^-_3}\]
Identify the conjugate acid-base pair for the following reaction in an aqueous solution.
\[\ce{NH^+_4 + CO^{2-}_3 ⇌ NH3 + HCO^-_3}\]
When aqueous ammonia is added to CuSO4 solution, the solution turns deep blue due to the formation of tetramminecopper (II) complex, \[\ce{[Cu(H2O)6]^{2+}_{( aq)} + 4NH3_{( aq)} ⇌ [Cu(NH3)4]^{2+}_{( aq)}}\], among HO2 and NH3 Which is stronger Lewis base.
Identify the conjugate acid - base pair in the following reaction.
\[\ce{H2O + HCl -> H3O+ + Cl-}\]
Which of the following species is amphoteric in nature?
The species which will behave both as conjugate acid and conjugate base is ______.
Define conjugate acid-base pair.
What are the limitations of Arrhenium theory?
