Advertisements
Advertisements
Question
Identify the optically active compounds from the following:
(i) \[\ce{[Co(en)3]^{3+}}\]
(ii) \[\ce{[trans - [Co(en)2Cl2]^+}\]
(iii) \[\ce{cis - [Co(en)2Cl2]^+}\]
(iv) \[\ce{[Cr(NH3)5Cl]}\]
Advertisements
Solution
(i) \[\ce{[Co(en)3]^{3+}}\]
(iii) \[\ce{cis - [Co(en)2Cl2]^+}\]
Explanation:
\[\ce{[Co(en)3]^{3+}}\] and \[\ce{[Co(en)2Cl2]^{2+}}\] are optically active compounds because their mirror images are non-superimposable isomer.

Non – superimposable isomers of \[\ce{[Co(en)3]^{3+}}\]
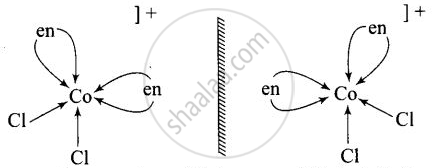
Non – superimosable isomers of \[\ce{[Co(en)2Cl2]^+}\]
APPEARS IN
RELATED QUESTIONS
Draw the geometrical isomers of complex \[\ce{[Co(en)2Cl2]+}\].
Indicate the types of isomerism exhibited by the following complex and draw the structures for these isomers:
[Co(en)3]Cl3
Indicate the types of isomerism exhibited by the following complex and draw the structure for this isomer:
[Pt(NH3)(H2O)Cl2]
How many geometrical isomers are possible in the following coordination entity?
[Cr(C2O4)3]3−
How many geometrical isomers are possible in the following coordination entity?
[Co(NH3)3Cl3]
Draw the structure of optical isomers of [Cr(C2O4)3]3−.
Draw the structure of optical isomers of [Cr(NH3)2Cl2(en)]+.
Draw all the isomers (geometrical and optical) of [CoCl2(en)2]+.
What type of structural isomers are [Co(NH3)5 Br] SO4 and [Co(NH3)5 SO4]Br? Give a chemical test to distinguish the isomers.
Name the type of isomerism exhibited by the following pairs of compound:
(1) (C2H5)2NH and CH3-NH-C3H7
(2) 1 – butanol and 2 methyl-1 -propanol.
Name the type of isomerism that the compound with molecular formula C3H6O2 exhibits. Represent the isomers.
Which one of the following complexes shows optical isomerism? (en = ethylenediamine)
Draw the geometrical isomers of [Co(en)2Cl2]2+. Which geometrical isomer of [Co(en)2Cl2]2+ is not optically active and why?
Indicate the types of isomerism exhibited by the following complexes and draw the structures for these isomers:
[Pt(NH3)(H2O)Cl2 ]
Assertion: Addition of bromine water to 1-butene gives two optical isomers.
Reason: The product formed contains two asymmetric carbon atoms.
Indicate the types of isomerism exhibited by the following complexes and draw the structure for these isomer:
\[\ce{[Pt(NH3)(H2O)Cl2]}\]
Indicate the types of isomerism exhibited by the following complex and draw the structure for this isomer:
\[\ce{[Pt(NH3)(H2O)Cl2]}\]
Indicate the type of isomerism exhibited by the following complex and draw the structure for the isomer:
\[\ce{[Pt(NH3 )(H2O)Cl2]}\]
