Advertisements
Advertisements
Question
Which one of the following complexes shows optical isomerism? (en = ethylenediamine)
Options
[Co(NH3)3Cl3]
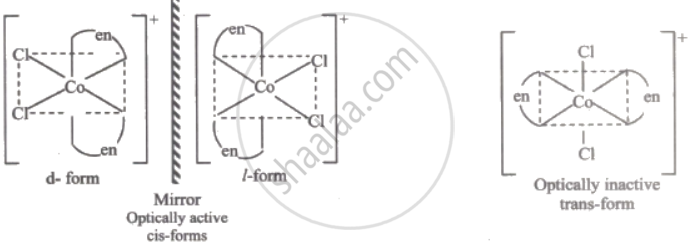
cis[Co(en)2Cl2]Cl
trans[Co(en)2Cl2]Cl
[Co(NH3)4Cl2]Cl
MCQ
Advertisements
Solution
cis[Co(en)2Cl2]Cl
Explanation:
- Optical isomerism occurs in chiral complexes that have non-superimposable mirror images.
- cis[Co(en)2Cl2]+ has two bidentate en ligands in a cis arrangement, making it chiral.
- The trans isomer and complexes with only monodentate ligands are achiral and do not show optical isomerism.
shaalaa.com
Isomerism in Coordination Compounds - Stereoisomerism
Is there an error in this question or solution?
