Advertisements
Advertisements
प्रश्न
Identify the optically active compounds from the following:
(i) \[\ce{[Co(en)3]^{3+}}\]
(ii) \[\ce{[trans - [Co(en)2Cl2]^+}\]
(iii) \[\ce{cis - [Co(en)2Cl2]^+}\]
(iv) \[\ce{[Cr(NH3)5Cl]}\]
Advertisements
उत्तर
(i) \[\ce{[Co(en)3]^{3+}}\]
(iii) \[\ce{cis - [Co(en)2Cl2]^+}\]
Explanation:
\[\ce{[Co(en)3]^{3+}}\] and \[\ce{[Co(en)2Cl2]^{2+}}\] are optically active compounds because their mirror images are non-superimposable isomer.

Non – superimposable isomers of \[\ce{[Co(en)3]^{3+}}\]
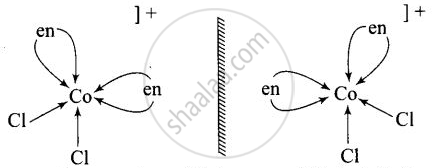
Non – superimosable isomers of \[\ce{[Co(en)2Cl2]^+}\]
APPEARS IN
संबंधित प्रश्न
Explain cationic complexes and anionic complexes of co-ordination compounds.
Draw one of the geometrical isomers of the complex [Pt (en)2Cl2] +2 which is optically inactive
Draw one of the geometrical isomers of the complex [Pt(en)2Cl2]2+ which is optically active.
What type of isomerism is exhibited by the complex [Co(en)3]3+?
(en = ethane-1,2-diamine)
Indicate the type of isomerism exhibited by the following complex and draw the structure for the isomer:
K[Cr(H2O)2(C2O4)2]
Indicate the type of isomerism exhibited by the following complex and draw the structure for the isomer:
[Co(NH3)5(NO2)](NO3)2
Draw the geometrical isomers of complex [Pt(en)2Cl2]2+.
What type of structural isomers are [Co(NH3)5 Br] SO4 and [Co(NH3)5 SO4]Br? Give a chemical test to distinguish the isomers.
Name the type of isomerism that the compound with molecular formula C3H6O2 exhibits. Represent the isomers.
Name the type of isomerism shown by the following pair of compounds:
[CoCl(H2O)(NH3)4]Cl2 and [CoCl2(NH3)4]Cl.H2O
Write the IUPAC name of [Co(en)2Cl2]+ ion.
The IUPAC name for [CoCl(NO2)(en)2]Cl is ____________.
Which of the following molecules has a chiral centre correctly labelled with an asterisk (*)?
Indicate the types of isomerism exhibited by the following complexes and draw the structure for isomers:
\[\ce{[Pt(NH3)(H2O)Cl2]}\]
Assertion: Addition of bromine water to 1-butene gives two optical isomers.
Reason: The product formed contains two asymmetric carbon atoms.
Indicate the types of isomerism exhibited by the following complexes and draw the structure for these isomer:
\[\ce{[Pt(NH3)(H2O)Cl2]}\]
Indicate the types of isomerism exhibited by the following complex and draw the structure for this isomer:
\[\ce{[Pt(NH3)(H2O)Cl2]}\]
Indicate the types of isomerism exhibited by the following complex and draw the structure for this isomer:
\[\ce{[Pt(NH3)(H2O)Cl2]}\]
