Advertisements
Advertisements
Question
How would you show that hydrogen is lighter than air?
Advertisements
Solution
Take a delivery tube and place one of its ends in a soap solution kept in a trough and the other one in a flat bottom jar as shown in the figure. The soap bubbles containing hydrogen rise upward the air. The rising soap bubbles prove that hydrogen is lighter than air.
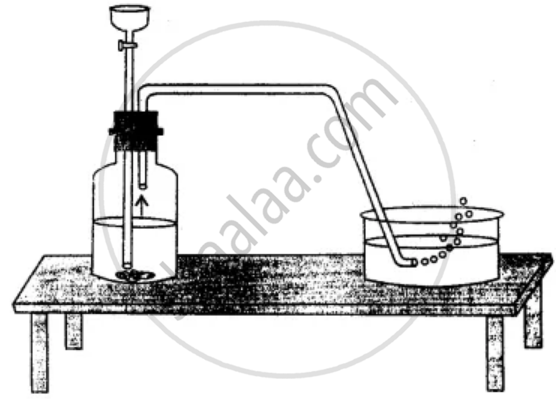
Hydrogen-filled soap bubbles rising upward in the soap solution and into the air shows that hydrogen is lighter than air.
APPEARS IN
RELATED QUESTIONS
Indicate which of the following statement is true and which is false:
Nitric acid cannot be used to prepare hydrogen by its action on active metals.
Name the following:
A metal which liberates hydrogen only when steam is passed over red hot metal.
Draw a neat and well-labelled diagram for the laboratory preparation of hydrogen.
Multiple Choice Question
In metal activity series the more reactive metals are at
FILL IN THE BLANK
Sodium liberates hydrogen when treated with cold .............
Give a test to identify hydrogen ?
Name the impurities present in hydrogen prepared in the laboratory.
How can these impurities be removed?
Under what conditions can hydrogen be made to combine with chlorine?
Name the products and write the equation for the reaction.
Give reason:
Apparatus for laboratory preparation of hydrogen should be airtight and away from a naked flame.
Starting from zinc how would you obtain hydrogen using Steam.
[Give balanced equation & name the product formed in the case other than hydrogen].
Name a metal which will not react with the reactants above to give hydrogen.
Starting from zinc how would you obtain hydrogen using a dilute acid.
[Give balanced equation & name the product formed in the case other than hydrogen].
Name a metal which will not react with the reactants above to give hydrogen.
Starting from zinc how would you obtain hydrogen using an alkali.
[Give balanced equation & name the product formed in the case other than hydrogen].
Name a metal which will not react with the reactants above to give hydrogen.
Give reason for the following:
Copper does not displace hydrogen from dilute hydrochloric acid, but zinc does.
Give a balanced equation for the following conversions sodium aluminate from aluminium.
In the laboratory preparation of hydrogen from zinc and dil. acid. Give a reason for the following:
Dilute nitric acid is not preferred as the reactant acid.
Select the correct answer from the symbol in the bracket.
The element which forms a diatomic molecule.
The diagram represent the preparation and collection of hydrogen by a standard laboratory method.

Name a gas other than hydrogen collected by the same method.
