Advertisements
Advertisements
प्रश्न
How would you show that hydrogen is lighter than air?
Advertisements
उत्तर
Take a delivery tube and place one of its ends in a soap solution kept in a trough and the other one in a flat bottom jar as shown in the figure. The soap bubbles containing hydrogen rise upward the air. The rising soap bubbles prove that hydrogen is lighter than air.
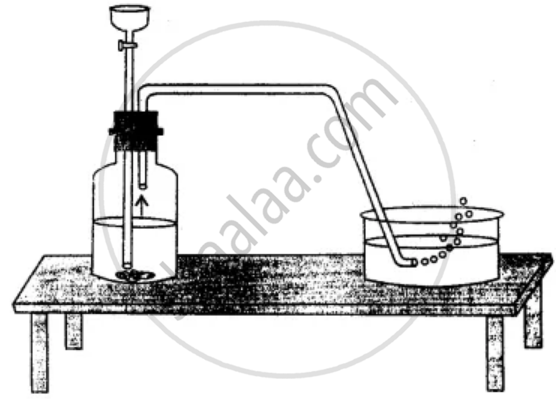
Hydrogen-filled soap bubbles rising upward in the soap solution and into the air shows that hydrogen is lighter than air.
APPEARS IN
संबंधित प्रश्न
Name the following:
A metal which liberates hydrogen only when steam is passed over red hot metal.
How is hydrogen gas collected? Why?
How would you show that hydrogen is a non-supporter of combustion?
Multiple Choice Question
In metal activity series the more reactive metals are at
FILL IN THE BLANK
Sodium liberates hydrogen when treated with cold .............
TRUE \ FALSE
Lead reacts briskly with dilute hydrochloric acid to form hydrogen.
Name the impurities present in hydrogen prepared in the laboratory.
Starting from zinc how would you obtain hydrogen using a dilute acid.
[Give balanced equation & name the product formed in the case other than hydrogen].
Name a metal which will not react with the reactants above to give hydrogen.
In the laboratory preparation of hydrogen from zinc & dilute hydrocholoric acid – state a reason for the collecting the hydrogen by downward displacement of water and not air & collecting it after all the air in the apparatus is allowed to escape.
Give balanced equation for the following conversion:
Zinc to sodium zincate – using an alkali.
Give balanced equation for the following conversion:
Acidified water to hydrogen – by electrolysis.
Give reason for the following:
Copper does not displace hydrogen from dilute hydrochloric acid, but zinc does.
Complete and balance the equation:
[Laboratory method]
By action of dilute acid on zinc
Zinc - Zn + 2HCl → _____ + _____ [g]
Give a balanced equation for the following conversions sodium aluminate from aluminium.
Name the following.
A gaseous reducing agent which is basic in nature.
Select the correct answer from the symbol in the bracket.
The element which forms a diatomic molecule.
The diagram represent the preparation and collection of hydrogen by a standard
laboratory method.

State what is added through the thistle funnel ‘Y’
The diagram represent the preparation and collection of hydrogen by a standard laboratory method.

State what difference will be seen if pure zinc is added in the distillation flask ‘X’ instead of granulated zinc.
