Advertisements
Advertisements
Question
Give reason for Sea water does not freeze at 0°C.
Advertisements
Solution
Sea water contains impurities that decrease its freezing point. That is why it does not freeze at 0°C.
APPEARS IN
RELATED QUESTIONS
Explain the following:
How can you relate the formation of water droplets on the outer surface of a bottle taken out of refrigerator with formation of dew?
On a winter day the temperature of the tap water is 20°C whereas the room temperature is 5°C. Water is stored in a tank of capacity 0.5 m3 for household use. If it were possible to use the heat liberated by the water to lift a 10 kg mass vertically, how high can it be lifted as the water comes to the room temperature? Take g = 10 m s−2.
Draw a graph between volume and temperature, when 5 cm3 of ice at -10°C is heated to form water at +10°C.
Explain, why are the exposed water pipes lagged with straw during severe winter?
Explain why do vegetables and fruits get damaged during severe frost?
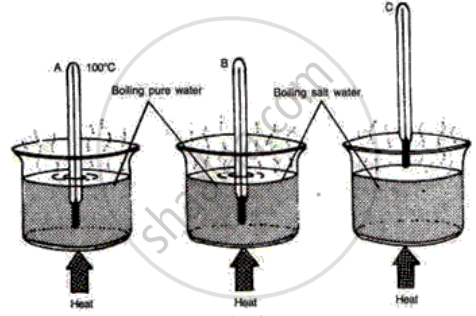
The following diagrams illustrate three situations involving thermometers which are labeled A, Band C. In each situation the thermometers indicate different readings.
(i) What do you expect the approximate reading of the thermometer B and C would be? Give a reason for your answer.
(ii) How would the readings of A and B help you in calibrating a thermometer?
In cold regions, in winter the pipes for water supply break.
